Molecular Docking Studies of Phytochemicals of Vitex Negundo (L) Against Adenosine A1 Receptor as Therapeutic Target in Cardiovascular Diseases
E. Maruthi Prasad1, Y. Nanda Kumar2, K. Lakshmi Devi1*
Affiliation
- 1Department of Biochemistry, Sri Krishnadevaraya University, Anantapuramu, India
- 2Biomedical Informatics Centre, Vector Control Research Centre, Indian Council of Medical Research, Pondicherry, India
Corresponding Author
Lakshmi Devi Kodidhela, Department of Biochemistry, Sri Krishnadevaraya University, Anantapuramu-515003, India. E-mail: klakshmidevibio@gmail.com
Citation
Kodidhela, L.D., et al. Molecular Docking Studies of Phytochemicals of Vitex Negundo (L.) Against Adenosine A1 Receptor as Therapeutic Target In Cardiovascular Diseases. (2015) Bioinfo Proteom Img Anal 1(2): 44- 48.
Copy rights
© 2015 Kodidhela, L.D. This is an Open access article distributed under the terms of Creative Commons Attribution 4.0 International License.
Keywords
5,3′-Dihydroxy-7,8,4′-trimethoxyflavanone; Adenosine A1 receptor; CVD; Molecular Docking
Abstract
Recent days, Ayurveda medicine is becoming one of the best alternatives for the modern medicines for effective control of Cardiovascular disease (CVD) due to their limited side effects and availability to a common man. Vitex negundo (L.) is a woody, large aromatic shrub growing to a small tree, which thrives in humid places or along water courses in wastelands, mixed open forests and found to occur widely in India, Afghanistan, Pakistan, Sri Lanka, Thailand, Malaysia, eastern Africa and Madagascar. The study evaluates a few selected phytochemicals (5,3′-dihydroxy-7,8,4′-trimethoxyflavanone; 5,3′-dihydroxy -6,7,4′trimethoxyflavanone; 5-hydroxy-7,4'dimethoxyflavone; 5,3'-dihydroxy-7,8,4'-trimethoxyflavanone; betulinic acid, ursolic acid; n-hentriacontanol; β-sitosterol) present in V. negundo plant leaves based on their capacity to bind and inhibit Adenosine A1 receptor of CVD using computational methods. Using the crystal structure of Adenosine A1 receptor from the Protein Data Bank, possible binding sites of Adenosine receptor were evaluated with CASTP server. Simultaneously, Molecular docking was performed using the GOLD (Genetic Optimization of Ligand Docking) software, to study the binding orientation of compounds to the Adenosine receptor. The efficiency and drug-likeness of various plant compounds were identified by using pre-ADMET software. In this study, all docked compounds were found to have interaction between an oxygen atom of the compounds and Adenosine receptor. In the binding pocket, common H-bonding interactions were formed between all docked compounds and GLY 135, GLN 137, GLN 140, HIS 143, GLU 145, GLN 145, GLN 207, ASP 209, HIS 232, THR 300, and HIS 311. The docking results agreed well with the observed in vitro data, which showed that the Adenosine receptor inhibitory activity of 5-hydroxy-7,4'dimethoxyflavone was higher than those of other compounds.
Introduction
Cardiovascular diseases (CVD) remain the principal cause of death in both developed and developing countries accounting roughly 20% of all deaths worldwide. It is estimated that 17.5 million people were died due to CVDs in 2012, representing 31% of global deaths. Among all these deaths, 7.4 million were because of coronary heart disease and 6.7 million were because of stroke[1,2]. Recent epidemiological survey indicated that urbanization and changes in life style are main causes for increase of CVDs. It is expected that by the year 2030, more than 23.3 million people will die annually due to CVDs. Asian countries are the home to 20% of the world’s population with the highest burden of CVDs in the world[3]. Myocardial Infarction occurs when the supply of blood to a part of the heart is interrupted. This is most commonly due to atherosclerotic plaque, which is an unstable collection of cholesterol and WBC (macrophages) in the wall of an artery[4]. The risk factors are family history, ethnicity and age, which cannot be controlled. Other risk factors that can be controlled include smoking, hypertension, obesity, high cholesterol, physical inactivity, type2 diabetes, unhealthy diets, oxidative stress, lipoproteins, marker enzymes and harmful use of alcohol etc[5,6].
It is well known that, Adenosine is a key endogenous molecule and potentially important signaling molecule in the heart.The adenosine production by cardiac myocytes reflects the metabolic state of the myocardium. Adenosine acts as regulator of vasodilation in the coronary resistance vessels, thereby coupling blood flow to the energetic needs of the heart[7]. It is well known that adenosine receptors are important therapeutic targets for cardiovascular diseases[8]. Adenosine regulates tissue function by activating four G-protein-coupled adenosine receptors: A1, A2A, A2B and A3; which control signaling pathways involved in regulating many body functions, especially in the cardiovascular system[9]. Activation of Adenosine A1 receptor (AAR) protects the heart from oxygen deficiency and slows down the heart rate, while activation of the A2 receptors can improve the flow of blood to the heart and lower blood pressure. These pathways are involved in inducing the CVDs leading to complications and push the life of an individual into the risk[10,11]. Hence, ceasing the activity of AAR in this metabolic pathway is suggested to be an ideal approach to prevent CVDs.
Natural compounds have been widely used for the discovery of many important modern drugs with novel mode ofaction[12]. V. negundo Linn. (Verbenaceae, Synonyms-Indian Privet; Nirgundi; Bana) is a woody, large aromatic shrub growing to a small tree. Different parts of V. negundo have cardiotonic, hepato-protective, antioxidant activity, anti-inflammatory, analgesic, cytotoxic activity, antihistamine property, antiulcerogenic, antiparasitic, antimicrobial, anti-fungal, antiviral activity and anti-arthritic potentials[13-23]. Leaves of V. negundo contains phytocompounds such as 5,3′-dihydroxy-7,8,4′-trimethoxyflavanone, 5,3′-dihydroxy -6,7,4′trimethoxy flavanone, 5- hydroxy-7,4’ dimethoxy flavones, 5, 3’- dihydroxy-7, 8,4’-trimethoxy flavanone, betulinic acid, ursolic acid, n-hentriacontanol and β-sitosterol[24] There exist no reports so far on the bioactivity of V. negundo with cardioprotective property and hence, in the present study we aimed to identify the cardio protective role of the phyto compounds of V. negundo through molecular modeling studies.
Methodology
Ligand Study
The leaves of V. negundo was reported to possess the active principles such as 5,3′-dihydroxy,6,7,4′ trimethoxyflavanone; 5,3′-dihydroxy-7,8,4′-trimethoxyflavanone; 5-hydroxy-7,4’dimethoxy flavone; betulinic acid; ursolic acid; n-hentriacontanol and β-sitosterol were 24 (Figure 1). We aimed to identify the inhibitory action of these compounds against AAR through molecular modeling studies. Initially the chemical structures of the compounds were retrieved from PubChem database and their three dimensional models were constructed in Discovery studio graphical environment and subjected to energy minimization in CHARMm force field to an RMS gradient of 0.01Å. The low energy and stabilized conformations of the compounds were saved and used for further studies.
Figure 1: The compounds known to be present in the leaves of V. negundo.
Sequence Retrieval and Template Identification
The protein sequence of AAR from Homo sapiens (ID: NP000665) was retrieved from NCBI and similarity search was carried out using BLAST-P server against PDB database. The hit with highest identity and coverage was taken as template to construct the homology model of AAR.
Homology Modeling of AAR
The homology model of the AAR was constructed using Modeller 9v11 software tool[25]. The Blast-P similarity search AAR against PDB revealed that a maximum identity of 51% was found with Human Adenosine A2 a Receptor that can be used as a template to construct the homology model of AAR. A sequence alignment file was generated between the template and AAR sequences and mentioned in python modeling script. The script file along with template structure was submitted to Modeller to run the script. A total of 20 models were generated and among themthe model with lowest DOPE (Discrete Optimized Potential Energy) was chosen as best model.
Protein Processing and Preparation through Molecular Dynamics Simulation
The structure constructed by using Modeller was optimized in Discovery studio software using CHARMm force field. The structure was energy minimized in 10,000 steps at a spherical cutoff of 12 Å for Vander Waals interactions and no periodic boundary conditions were added providing the implicit solvent environment. The energy minimized structure of AAR was then prepared using protein preparation wizard to clean up the common internal errors of the structure. Loops were defined with a maximum length of 20 residues with subsequent CHARMm minimization. The structure is then protonated with a protein dielectric constant of 10, pH 7, ionic strength of 0.145 M and with an energy cutoff of 0.9 kcal/mol. The protonated structure was subjected to molecular dynamics simulations using standard dynamics cascade. The simulations were started with a primary minimization step using Steepest Descent algorithm to improve the poor conformational regions and followed by secondary energy minimization step using Conjugate Gradient algorithm to an RMS gradient of 0.1Å in a total of 2000 steps. The system was heated from 30 K to 300 K in a time steps of 100 pico seconds (ps) with a velocity frequency of 50 steps. Further the system was equilibrated in 100 ps and finally followed by production phase of 10000 ps. The generated conformations were studied for their total energies and RMSD to look into the stability of the system. The low energy conformation obtained at the end of the simulation was trapped from the total trajectories and used for the further studies.
Structure Validation
The stereo chemical quality of the final structure was analyzed by Ramachandran plot using PROCHECK validation server[26] and Z-Score plot of ProsaWeb server[27].
Binding Site Identification
CASTp online server was employed to predict the possible binding sites on the AAR structure. CASTp identifies and measures pockets and pocket mouth openings, as well as cavities. This program specifies the atoms lining pockets, volume and area of pockets and cavities; pocket openings, and buried cavities; and the area and circumference of mouth openings[28]. Further the functional patterns were also predicted by submitting the AAR protein sequence to PROSITE secondary database[29]. The idea behind the usage of these two servers is the correlation of results from both sources will help to predict more reliable binding site that can be targeted with the compounds.
Molecular Docking Studies
Docking studies were carried out using a high-throughput algorithm, LibDock of Discovery Studio. The energy minimized compounds were docked into the predicted binding site of AAR homology model with an XYZ coordinates of -51.24, -1.47 and -33.11 respectively. Ligand conformations were aligned with polar and non-polar receptor sites which are called as hotspots. A maximum of 100 hot spots were defined with a docking tolerance of 0.25 Å to accept or reject any specific conformation. BEST conformation method was applied to ensure the maximum conformational space of the ligand in the binding site. Multiple docking conformations were generated for each ligand and evaluated by LibDock scores. The docked conformations were subsequently energy minimized to an RMSD cutoff of 1Å in 1000 maximum steps. Energetically, the most favorable conformation was selected and their intermolecular interactions in the active site of AAR were analyzed in the best energy docked conformations[30].
Results and Discussion
Homology Modeling of AAR
A high percentage of sequence similarity should be more accurate alignment between the target sequence and template structure. In our study, we have chosen crystal structure of human adenosine A2a receptor as a reference structure for constructing AAR model (PDB ID: 3VG9). A total of 20 modHomology modeling of AAR A high percentage of sequence similarity should be more accurate alignment between the target sequence and template structure. In our study, we have chosen crystal structure of human adenosine A2a receptor as a reference structure for constructing AAR model (PDB ID: 3VG9). A total of 20 models were generated for AAR and among the model with lowest DOPE (Discrete Optimized Potential Energy) score was chosen.
Molecular Dynamics Simulations
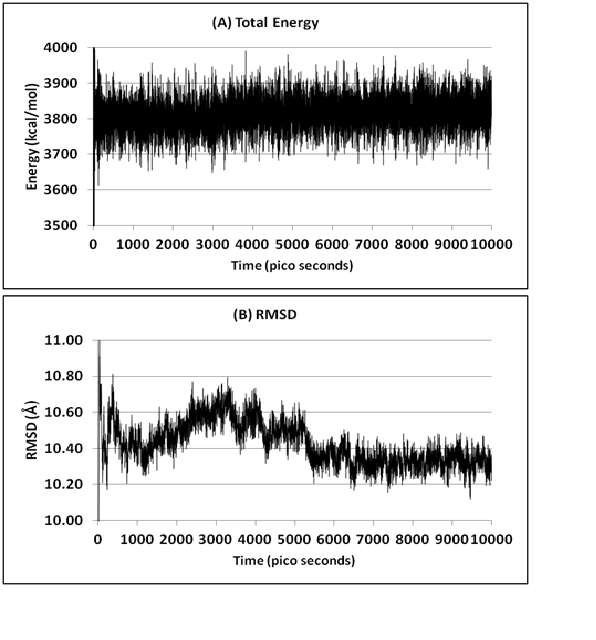
The constructed homology model was energy minimized and protonated in Discovery Studio software followed by molecular dynamics simulations to get the stabilized conformation. The molecular dynamics simulations using standard dynamics cascade generated around 20000 trajectories in 10000 ps of production time. All these trajectories were analyzed based on their energy levels and RMSD values. The total energy plot revealed that the AAR homology was stabilized at the energy levels of 3800 kcal/mol and no fluctuations were observed by the end of simulation period (Figure 2A). Further, the RMSD plot revealed that the model was fluctuated for a period of 8000 ps and later onwards stabilized at an RMSD range of 10.3 Å (Figure 2B). From these observations, the stabilized trajectory was trapped at the end of simulation period. In addition, these simulation results explain the overall stability of the AAR homology model and suggest its use for further studies.
Figure 2: (A) Total energy and (B) RMSD plots of AAR in 10000 ps molecular dynamics simulations.
Structure Validation
The validation of the protein model was carried by using Ramachandran plot (Figure 3A). The plot statistics showed that 94.8% of the residues were in most favored regions, 4.5% in additional allowed regions, 0.7% in generously allowed regions and no residues were observed in disallowed region. This explains the reliable validity of the models with acceptable stereo chemical quality. Further ProsaWeb plot (Figure 3B) stated that, it showed a Z-score of -3.96 which has fallen in the range of X-ray crystallographic regions indicating the near native nature of the homology model. These two validations are strong enough to proceed for further analysis.
Figure 3: (A) Ramachandran plot showing the stereo chemical quality of the AAR model. Glycine residues are shown as triangle, Proline residues as square and remaining residues as circles. (B) ProsaWeb plot of AAR homology model showing a Z-score of -3.96 falling in the range of native X-ray crystallographic region.
Prediction of Binding Site
Multiple binding sites were predicted in the stabilized model of AAR using CASTp server. PROSITE results revealed that the AAR protein contains G-protein coupled receptors family 1 signature ranging from 93-109 residues. These residues were in correlation with the CASTp results and hence the same residues such as Ser93, Ser94, Ile95, Leu96, Ala97, Leu98, Leu99, Ala100, Ile101, Ala102, Val103, Asp104, Arg105, Tyr106, Leu107, Arg 108 and Val109 were chosen as binding site. These residues were also observed to contain a perfect binding cavity conformation in the AAR structure and hence chosen as the more favorable sites to dock the ligands (Figure 4).
Figure 4: Homology model of AAR showing the predicted binding site. The AAR structure is represented in cartoon model and the binding site in sphere surface. Left: lateral view; Right: Top view. The binding site is confined to the pore region of the model.
Molecular Docking
The seven compounds were successfully docked into the predicted binding cavity of AAR model using LibDock module. Multiple conformations were generated for each compound and the conformations were ranked using LibDock score. The observation of LibDock score revealed that Lig1 showed a highest docking score of 111.36 kcal/mol and the lowest docking score of 70.47 kcal/mol was observed for Lig5 (Table 1). According to LibDock algorithm, higher the docking score higher the strength and lower the docking score lower the strength of the docking complex. So, it can be inferred from the docking scores that, Lig1 is having most binding affinity among all the compounds and could best inhibit the AAR activity.
Table 1: Molecular docking of Phytochemicals of Vitex negundo (L.) against human AAR protein structure.
| Ligand | Name | LibDock Score (kcal/mol) |
| Lig1 | 5,3′-dihydroxy-7,8,4′-trimethoxyflavanone | 111.36 |
| Lig2 | 5,3′-dihydroxy -6,7,4′-trimethoxyflavanone | 90.84 |
| Lig3 | 5-hydroxy-7,4’-dimethoxyflavone | 110.20 |
| Lig4 | Betulinic acid | 80.15 |
| Lig5 | Ursolic acid | 70.47 |
| Lig6 | n-hentriacontanol | 70.83 |
| Lig7 | β-sitosterol | 90.48 |
These compounds were observed to interact with the AAR binding site by means of hydrogen bonding and hydrophobic interactions. Lig1 was observed to form hydrogen bonding with Ala43, Thr44 and Arg105 residues using its oxygen reactive centres. Such kinds of hydrogen bond interactions were also observed in Lig2, Lig3 and Lig7. The residue, Arg123 of AAR was observed to play a vital role to interact with the ligands by means of hydrogen bonding. Hydrophobic interactions were observed with Lig4, Lig5 and Lig6, which were favored by their aromatic rings (Figure 5). The hydrophobic resides such as Ala100, Val103, Tyr106, Val118, Val119 and Ala127 were observed to form hydrophobic spheres with the ligands. Hydrophobic interactions represent more stable interactions than hydrogen bonds. The compounds, which did not formed hydrogen bonds, were stabilized by hydrophobic interactions by orienting towards the hydrophobic amino acid residues in the AAR binding site. Although there exists variable docking scores among seven phytochemicals, the orientation of the compounds was observed to fit snugly in the binding site favoring their strong binding with AAR. Such a molecular interactions and affinity levels of these seven phytochemicals explains their inhibitory action against AAR protein. These results will finally suggest their use as AAR inhibitors and further their in vitro evaluation is needed to be ascertained.
Figure 5: Molecular interactions of phytochemicals of Vitex negundo (L.) in the binding site of AAR model. The ligands are shown in CPK colors and the AAR amino acid residues are shown in orange color stick models.
Conclusion
In the present study we aimed to identify the phytochemicals of V. negundo (L.) as AAR protein inhibitors so as to use for the control and management of CVDs. As a preliminary study, we have modeled the AAR protein structure by homology modeling method and stereo chemical quality was validated. A stabilized trajectory of the model was obtained by molecular dynamics simulations and a potential binding site was predicted using CASTp and PROSITE servers. Molecular docking studies of the seven phytochemicals using LibDock module revealed their potential (5,3′-dihydroxy-7,8,4′-trimethoxyflavanone) to inhibit AAR protein and in turn suggesting their use for the treatment and management of CVDs.
References
- 1. Global status report on non-communicable diseases. (2014).
- 2. Panda, V.S., Desai, Y.H., Sudhamani, S. Protective effects of Macrotyloma uniflorum seeds (horse gram) in abnormalities associated with the metabolic syndrome in rats. (2015) J Dia Obes 2(2): 1-10.
- 3. Mathers, C.D., Loncar, D. Projections of global mortality and burden of disease from 2002 to 2030. (2006) PLoS Med 3(11): e442.
- 4. Shaik, A.H., Rasool, S.N., Reddy, V.K., et al. Cardioprotective effect of HPLC standardized ethanolic extract of Terminalia pallida fruits against isoproterenol-induced myocardial infarction in albino rats. (2012) J Ethnopharmacol 141(1): 33- 40.
- 5. Ralph, L.S., Emelia, J. B., Joseph, P. B., et al. Risk Factors. (1997) Stroke 28: 1507-1517.
- 6. Liu, H., Jabir, B. Association between Ethanol Consumption and Hypertension Risk. (2015) J Heart Cardiol 1(2): 1-4.
- 7. Chen, Y.J., Bache, R.J. Adenosine: A Modulator of the Cardiac Response to Stress. (2003) Circ Res 93(8): 691-693.
- 8. Chen, J.F., Eltzschiq, H.K., Fredholm, B.B. Adenosine receptors as drug targets- what are the challenges? (2013) Nat Rev Drug Discov 12(4): 265-286.
- 9. Sheth, S., Brito, R., Mukherjea, D., et al. Adenosine Receptors: Expression, Function and Regulation. (2014) Int J Mol Sci 15(2): 2024-2052.
- 10. Mustafa, S.J., Morrison, R.R., Teng, B., et al. Adenosine Receptors and the Heart: Role in Regulation of Coronary Blood Flow and Cardiac Electrophysiology. (2009) Handb Exp Pharmacol 193: 161-188.
- 11. Kaiser, S.M., Quinn, R.J. Adenosine receptors as potential therapeutic targets. (1999) Drug Discov Today 4(12): 542-551.
- 12. Ji, H.F., Li, X.J., Zhang, H.Y. Natural products and drug discovery. Can thousands of years of ancient medical knowledge lead us to new and powerful drug combinations in the fight against cancer and dementia? (2009) EMBO Rep 10(3): 194-200.
- 13. Vishwanathan, A. S., Basavaraju, R. A Review on Vitex negundo (L.)- A Medicinally Important Plant. (2010) EJBS 3(1): 30-42.
- 14. Pai, P.T., Adnaik, R.S., Mule, S.N., et al. Evaluation of cardiotonic activity of leaves of Vitex negundo Linn. (2009) Int J Green Pharm 3(4): 306-309.
- 15. De, S., Ravishankar, B., Bhavsar, G.C. Plants with hepatoprotective activity: A review. (1993) Ind Drugs 30(8): 355-363.
- 16. Dharmasiri, M.G., Jayakody, J.R., Galhena, G., et al. Anti-inflammatory and analgesic activities of mature fresh leaves of Vitex negundo. (2003) J Ethnopharmacol 87(2-3): 199-206.
- 17. Telang, R. S., Chatterjee, S., Varshneya, C. Study on analgesic and anti-inflammatory activities of Vitex negundo linn. (1999) Indian J of Pharmacol 31(5): 363-366.
- 18. Chowdhury, J.A., Islam, M.S., Asifuzzaman, S.k., et al. Antibacterial and cytotoxic activity screening of leaf extracts of Vitex negundo (Fam: Verbenaceae). (2009) J Pharm Sci & Res 1(4): 103-108.
- 19. Sahni, Y.P., Srivastava, D.N., Gaidhani, S.N. Protective activity of some indigenous medicinal plants on gastric ulceration in albino rats (2001). Journal of Medicinal and Aromatic Plant Sciences 22/23(4A/ 1A): 89-90.
- 20. Kamaraj, C., Rahuman, A.A., Bagavan, A., et al. Evaluation of medicinal plant extracts against blood-sucking parasites. (2010) Parasitol Res 106(6): 1403-1412.
- 21. Rusia, K., Srivastava, S. K. Antimicrobial activity of some Indian medicinal plants. (1998) Ind J Pharm Sci 50: 57-58.
- 22. Parveen, N. Antifilarial activity of Vitex negundo against Setariacervi. (1991) Fitoterapia 62(2): 163-165.
- 23. Saravana, K. K., Nagaveni, P., Mahaboob, S. T. Evaluation of Anti-arthritic activity of Vitex Negundo Linn. (2013) International Journal of Research in Pharmacy and Life Sciences 1(1): 1-4.
- 24. Ladda, P. L., Magdum, C. S. Vitex negundo (L.) Ethnobotany, Phytochemistry and Pharmacology- A Review. (2012) IJAPBC 1(1): 111-120.
- 25. Mohammed, S.A., Manjunatha, S., Dase, G.K.R., et al. Homology modelling and docking studies of phytocompounds from Trigonella foenum graecum for anti diabetic activity. (2015) WJPR 4(8): 1120-1130.
- 26. Prasad, E.M., Ibrahim, A.S., Devi, K.L., et al. Drug designing and docking studies of breast cancer type1 susceptibility protein (brca1) involved in breast cancer. (2014) Int J Pl An and Env Sci 4(2): 752-763.
- 27. Kumar, P.S., Kumar, Y.N., Prasad, U.V., et al. Comparative structural and functional analysis of Staphylococcus aureus glucokinase with other bacterial glucokinases. (2014) Indian J Pharm Sci 76(5): 430-436.
- 28. Paramashivam, S.K., Elayaperumal, K., Natarajan, B.B., et al. In silico pharmacokinetic and molecular docking studies of small molecules derived from Indigofera aspalathoides Vahl targeting receptor tyrosine kinases. (2015) Bioinformation 11(2): 73-84.
- 29. Sigrist, C.J., De Castro, E., Cerutti, L., et al. New and continuing developments at PROSITE. (2013) Nucleic Acids Res 41: D344-347.
- 30. Kumar, Y.N., Jeyakodi, G ., Thulasibabu, R., et al. Screening and identification of inhibitors against glutathione synthetase, A potential drug target of Plasmodium falciparum. (2015) Comb Chem High Throughput Screen 18(5): 492-504.