Total Antioxidant Capacity, Antimicrobial Activity and Preliminary Analysis of Some Nutritional Compounds in Moringa Oleifera preparations
Enrico Doria1,3*, Bakari Daoudou1,2, Daniela Buonocore3, Manuela Verri3, Maurizia Dossena3, Lethabo Mashigo1, Steven Manzi4 and Abdulkadir A Egal1
Affiliation
- 1Centre of Sustainable Livelihoods, Vaal University of Technology, Vanderbijlpark, South Africa
- 2Department of Biological Science, Faculty of Sciences, University of Maroua, Maroua, Cameroon
- 3Department of Biology and Biotechnology, University of Pavia, Italy
- 4Department of Biotechnology, Vaal University of Technology, Vanderbijlpark, South Africa
Corresponding Author
Enrico Doria1, Centre of Sustainable Livelihoods (CSL), Vaal University of Technology, EF04, Science Park, Private Bag X021, Andriers Potgieter Boulevard, Vanderbijlpark, 1900, South Africa; Tel: + 39-0382985580; E-mail: enrico.doria@unipv.it
Citation
Doria, E., et al. Total Antioxidant Capacity, Antimicrobial Activity and Preliminary Analysis of Some Nutritional Compounds in Moringa oleifera preparations. (2017) Int J Food Nutr Sci 4(1): 23- 29.
Copy rights
© 2016 Doria, E. This is an Open access article distributed under the terms of Creative Commons Attribution 4.0 International License.
Keywords
Antioxidant activity; Flavonoids; Functional food; M. oleifera; Phytic acid, Trigonelline, Ursolic acid
Abstract
Globally, there is a growing interest in plant products as a potential source of antioxidant compounds in relation to oxidative stress that is one of the causes of several degenerative diseases. Moringa oleifera is one of the African plants commonly consumed as a nutritional supplement by several communities, due to the high level of vitamins and minerals present in the leaves. In this study, the antioxidant power, some nutritional (polyphenols, vitamin C, flavonoids as kaempferol and quercetin, ursolic acid and trigonelline) and anti nutritional factors (condensed tannins, phytic acid) from a leaf powder extract, obtained from a Moringa cultivar grown in Guateng region (South Africa), were tested. Some of these results were compared to a commercial Rooibos tea (Aspalathus linearis), another typical and commonly used South African beverage, and to coffee (Coffee arabica). The results of these comparison tests indicated that tea preparations from Moringa oleifera powder and from Rooibos tea contains the same antioxidant activity but the content of flavonoids, vitamin C and free phosphate are significantly higher in Moringa. Moreover, Moringa extract shows antimicrobial and antifungal activity and very significant concentrations of ursolic acid (first evidence in moringa) and trigonelline
Introduction
Moringa oleifera, a tree native of northern India, is an exceptionally nutritious plant with a variety of potential uses and widely cultivated throughout the tropics[1]. It is known to be extremely valuable in local communities where most of the people consume plant-based diets[2]. Almost all the parts of this plant, from seeds to the roots, are used as various ailments in the indigenous medicine system, whereby the leaves possess remarkable nutritional and medicinal qualities[3,4]. The leaves of Moringa oleifera contain significantly amounts of vitamin C, A, and E, carotenoids, minerals (calcium, potassium and iron mostly), proteins, essential amino acids as well as antifungal, antiviral and anti-inflammatory agents[5,6]. All above-mentioned nutritional and supplement properties of Moringa oleifera could play an important role in the fight against malnutrition in both developing and under developed countries[3]. Local consumption of Moringa is also increasing as a consequence of rising publicity about its health benefits, especially regarding the diabetics, obesity and fever treatment[7,8]. Nowadays, many companies across the world are manufacturing various products containing Moringa leaves in form of tablets, capsules, leaf powder or tea containing a relevant amount of antioxidant compounds helping to reduce risk of cardiovascular diseases and decreasing also the cholesterol level[9,10,11]. However, several differences with regards to methods used for the tea preparation in the stages of maturation of the leaves after harvesting, the type of tree species and environmental differences of the regions where the tree is cultivated, have to be taken into account[12]. Moreover, some studies have showed an interesting level of flavonoids and polyphenols in Moringa leaves; these secondary metabolites, due to their important antioxidant property, are involved in anti-carcinogenic, anti-viral, anti-estrogenic and immune-stimulating activities[13,14,15,16,17]. For these reasons, currently, there is a growing interest in the use of Moringa leaf as an ingredient in the preparation of herb tea, considered as functional foods along with beverages such as sports drinks, fruit and vegetable juices[11,18]. In the developed countries, where the nutritional problems are less prevalent, commercial preparation of Moringa oleifera is used in sport activities to enhance physical energy and athletic performance.
In this study we measured the antioxidant properties, antimicrobial activity and the level of some compound with nutritional (polyphenols, flavonoids) or anti-nutritional activity (phytic acid, condensed tannins) of extracts of Moringa oleifera species cultivated in South Africa and we compared with other commonly used hot drinks as rooibos tea and coffee. Furthermore some other interesting compounds like ursolic acid and trigonelline, present in the Moringa leaves, were analyzed. Ursolic acid is a terpenoid compound involved in the skeletal muscle and energy expenditure increasing, improving glucose tolerance and decreasing the hepatic steatosis. Therefore ursolic acid is a potential therapeutic approach for obesity and obesity- related illness[19]. Trigonelline is an alkaloid with therapeutic potential for diabetes and central nervous system disease[20] and possess an interesting phytoestrogenic activity[21].
Materials and Methods
Plant material: Moringa oleifera leaves (6 months old) were obtained from Green Hearth®, in Gauteng region, South Africa. Rooibos tea and coffee commercial preparations were bought in a local shop.
Chemicals and solvents: All reagents and standard chemicals were purchased from Sigma Aldrich®.
Samples preparation: Samples for total antioxidant capacity (DPPH test) and total polyphenols analysis were treated according to the method described by[22]. An amount of 200 mg of samples (in triplicate) was extracted with 40 ml of methanol: water (50:50, v/v; pH 2.0) and homogenized for 5 minutes at room temperature. The homogenate samples were kept at 4°C for 1 h and then centrifuged at 2500 g for 10 minutes. The supernatants were recovered and the residue was further washed with 40 ml of acetone: water (70:30, v/v) solution and then centrifuged. The resulting supernatants were combined and stored at 20°C before analysing.
Total antioxidant capacity (DPPH test)
By means of the widely used 2,2-Diphenyl-1-Picrylhydrazyl (DPPH) test, following the procedure described by Brand-Williams et al.,1995[23], modified by Thaiponga et al., 1995[24], it is possible to measure the ARP (Anti-Radical Power) of extracts prepared from any biological material. Stock solution was prepared by dissolving 24 mg of DPPH powder in 100 ml of methanol and then stored at 20°C until needed. The working solution was obtained by mixing 10 ml of stock solution with methanol: water 1:1 (v/v) to obtain an absorbance of around 1.0 ± 0.1 units at 515 nm (UV/VIS Perkin-Elmer Lambda 35 spectrophotometer). Different amounts (10 – 100 μl) of prepared samples were added (except in the control samples) to 900 μl of DPPH working solution. This stable free radical reacts with antioxidants and its consequent colour loss, measurable at 515 nm, correlates with antioxidant content. Each reaction mixture was incubated overnight at room temperature in the dark so as to let it reach a steady state before the final absorbance was read and the residual concentration of DPPH calculated by making reference to a calibration curve (slope: 0.029) obtained by measuring absorbance at 515 nm of a series of dilutions in methanol: water (1:1) of a freshly prepared DPPH methanolic solution (1 mg/ml), so as to span the range 0.1 - 100 mg/ml. These values were plotted against those of the volumes (ml) of extract added at time zero to the DPPH solution in order to calculate by interpolation the volume of extract required to consume 50% of the initial DPPH amount. The reciprocal of this figure corresponds to the ARP value[25].
Determination of total polyphenolic content
Total phenolics were determined by the method described by Waterman, Mole 1994[26], using Folin–Ciocalteu reagent. In a test tube, 150 μl of the methanol–acetone extract, 2400 μl of nano pure water and 150 μl of 0.25 N Folin–Ciocalteu reagent were combined and then well mixed, using a vortex. The mixture was allowed to react for 3 minutes after which 300 μl of 1N Na2CO3 solution was added and mixed well. The solution was incubated at room temperature for 2 hours and the absorbance was measured at 725 nm against a blank. Quantification was based on the standard curve of Gallic acid solution (0 – 0.5 mg/ml) and results were expressed as mg/g of GAE (gallic acid equivalents)[27].
HPLC determination of quercetin and kaempferol
Flavonoids were extracted and assayed according to the modified method described by Gudej et al., 2004[28]. Briefly, 200 mg of material (three replicates) were extracted with 6 ml of 25% HCl and 20 ml of methanol. The samples were then heated and shaken at 85°C for 2 hours for glycosides hydrolyzation. A fraction (5 ml) of the treated samples was then transferred in a new flask where 10 ml of methanol were added. The samples were then centrifuged for 10 min at 10,000 g and filtered (0.45 nm) before to be analysed by HPLC system (Perkin Elmer series 200 equipped with diode array detector UV/Vis and a C18 column, Mediterranean 5 μm 25 × 0.46). The solvents were (A) methanol and (B) water adjusted to pH 2.8 with acetic acid. The injection volume was 20 μl and the flow rate 1 ml/min. The elution profile was as follows: 0 min. 40% A in B, 0 - 0.5 min 40% to 60% A in B, 0.5 - 2.5 min 65 A in B, 2.5 - 6.0 min 6 to 45% A, 6.0 - 8.0 min 40% A in B. UV absorbance at 260 nm was used to detect quercetin and kaempferol in the same chromatographic run. flavonols were quantified by comparing the peak height in the chromatograms with the value registered in the calibration curve obtained for each compound by injecting 20 μl of different dilutions of the respective standards dissolved in dimethyl sulfoxide (DMSO).
Condensed tannins extraction and analysis
An amount of 0.5 g (either for Moringa leaf or Rooibos powder) was mixed with 10 ml of acetone: water: diethyl ether (7 : 2 : 1) solution and shaken at 60°C for 1 hour in the dark. The samples (in triplicate) were then sonicated and centrifuged at 8000 g for 10 minutes (4°C) and the supernatant was filtered in new test tubes. An aliquot (0.5 ml) of each extract was mixed with 3 ml of butanol/HCl (95:5, v/v) solution in screw-capped test tubes and incubated for 60 minutes at 95°C. A red coloration was developed and the absorbance was then read at 550 nm versus a prepared blank and compared with a known concentration range of delphinidin standard, similarly prepared. All results were expressed as mg delphinidin equivalents/g dry material. Data are reported as mean ± standard deviation for three replications. A linear response was obtained between 1 μg and 5 μg of delphinidin/ml solution[26].
Determination of phytic acid and free phosphate
In order to measure the free phosphate content, a flour sample of 50 mg from each plant material (3 replicates) was extracted with 1 ml of 12.5% TCA, 25 mM MgCl2 solution for 20 minutes at room temperature and then left stirring overnight at 4°C. After centrifugation, 100 ml of the supernatant were added to 900 ml of a freshly prepared Chen’s reagent (6N H2SO4 : 2.5% ammonium molybdate : 10% ascorbic acid : H2O (1:1:1:2, v/v/v/v)) and incubated at 50°C for 1 hour before reading the absorbance at 650 nm of the blue reaction mixture[29]. A reference standard curve was routinely prepared using a series of Na2HPO4 solutions within the linearity range (from 10 to 60 nanomoles of phosphate). Phytic acid content was determined in a similar way after samples were subjected to a ferric precipitation method, as described by Pilu et al., 2005[30] and expressed as mg/g. Briefly, after precipitation with Iron chloride solution, the extracts were incubated at 100°C for 30 minutes; after centrifugation, the supernatant was discarded and pellet suspended twice using 400 μl of 0.2 M HCl and a third time using 400 μl of concentrated H2SO4. The sample mixture was left at 100°C adding day by day and continuatively small amount of 3% H2O2 until the samples color is completely cleared. After 24 h without adding hydrogen peroxide, the samples were assayed using the Chen’s reagent.
Determination and extraction of Vitamin C
Vitamin C from Rooibos and Moringa leaf powder was determined by using the HPLC system previously described. An amount of 0.2 g of plant material (in triplicate for each plant species) was extracted with 70% methanol containing 3% H3PO4. After shaking for 30 minutes at room temperature, the samples were sonicated and filtered before injecting in HPLC. The isocratic mobile phase was 2% KH2PO4 (pH 2.3); the flow rate was 0.8 ml/min, and samples were run for 15 minutes with a post-run period of 2 minutes. The detection was performed at 254 nm; L-Ascorbic acid was identified and quantified by comparing retention times, absorption spectra, and peak areas with those of L-ascorbic acid standard[31].
Trigonelline and ursolic acid determination
Samples of two grams (in triplicate) from Rooibos and Moringa leaf powder were extracted for 1 hour at room temperature with methanol containing 1% 2,6-di-tert -butyl-4-methylphenol (BHT) using an ultrasonic bath[32]. The samples were then filtered through a membrane filter (0.45 μm pore size) prior to injection. The HPLC method was set up to allow the simultaneous determination of the two compounds. Mobile phases were methanol (A) and 0.01M H3PO4 (B); flow rate was 0.6 ml/min, and samples were run with 90 of A for 15 minutes with a post-run period of 5 minutes with 95% of A phase. The detection was performed at 210 nm and the samples were quantified by comparing the peak height in the chromatograms with the value registered in the calibration curve obtained for each compound by injecting different dilutions of the standards dissolved in methanol[33].
Determination of antimicrobial activity
List of the tested microorganism:
• Enterococcus faecalis (ATCC 29212) (Gram positive)
• Klebsiella pneumoniae (ATCC 9633) (Gram negative)
• Pseudomonas aeruginosa (ATCC 15442) (Gram negative)
• Staphylococcus aureus (ATCC 25912) (Gram positive)
• Cryptococcus neoformans (ATCC 14116) (Yeast)
• Candida albicans (ATCC 14053) (Yeast)
Antibacterial activity
Minimum inhibitory concentrations (MIC) of Moringa extracts for antibacterial activity were determined using micro dilution bioassay[34]. Cultures of gram positive (Enterococcus faecalis ATCC 29212) and gram negative (Klebsiella pneumoniae ATCC 9633) were diluted with sterile Mueller-Hilton broth to obtain final inoculums of approximately 106 CFU/ml (colony forming units). Moringa powder extracts were suspended in acetone to a concentration of 100 mg/ml. One hundred micro liters of each extract was serially diluted with sterile distilled water in a 96 well microtiter plate for each of the four bacterial strains. Serial dilutions of neomycin (starting from 0.1 mg/ml) were used as a positive control. Each strain culture was then incubated at 37 for 24 h. Bacterial growth (MIC) was monitored after addition of 50 μl of 0.2 mg/ml of the dye indicator INT (piodo nitro tetrazolium chloride) and further incubation at 37°C for 24h[34].
Antifungal activity
Micro dilution method described by Masoko, Eloff 2007[34,35] was used to determine antifungal activity of Moringa extracts. The used protocol is the same previously described for antibacterial activity with exception of the amphotericin B antibiotic as positive control. Moreover the incubation period of the fungal strain was 48 hours.
Results and Discussion
Antioxidant activity and polyphenols analysis
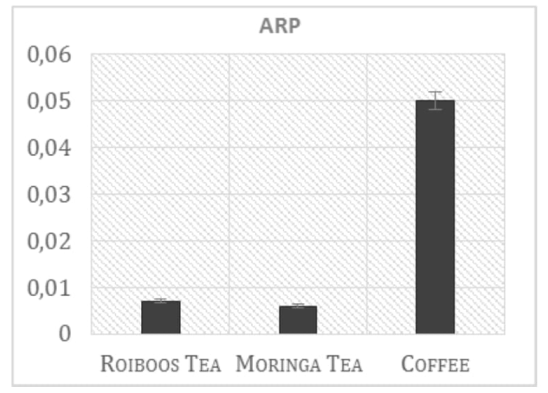
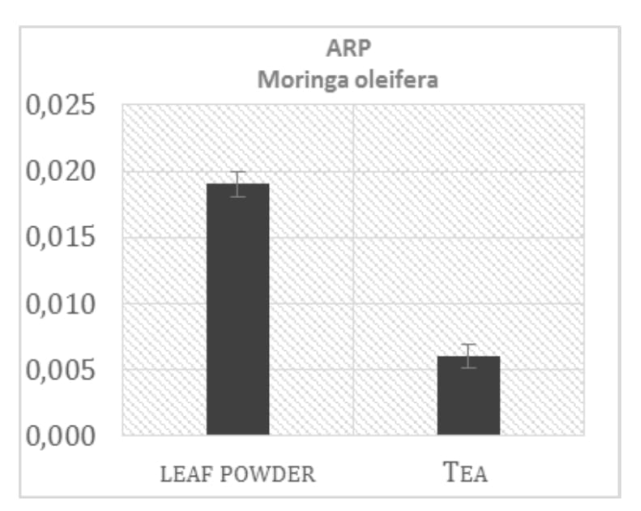
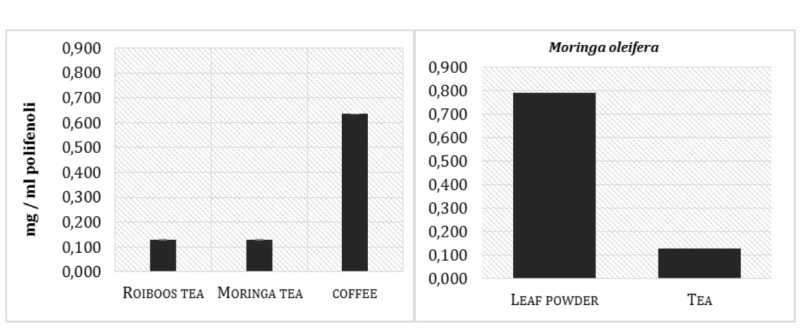
In order to collect information about the level of overall antioxidant activity present in Moringa leaf extract, the DPPH test was used. Even not a biological radical, DPPH test provides a good estimation of the total antioxidant activity in the examined samples. Results are presented in the Figure 1 and show the ARP values from Moringa tea extract (infusion prepared by using of dry leaves) compared with the other two commonly used beverages in South Africa, Rooibos tea and coffee (mixed coffee varieties, available in the stores). Coffee drink presents, as expected, the higher total antioxidant capacity (0.05 of ARP, antiradical power), around eight fold more than Moringa and Rooibos tea (0.007 and 0.006 respectively). Moringa tea preparation presents the same antioxidant activity showed by the Rooibos tea, but comparing the ARP values obtained from the leaf powder and from tea preparation, it was possible to notice a relevant difference, as shown in the Figure 2. Moringa leaf powder, in fact, presents an antioxidant activity more than three times higher respect to that found in tea infusion preparation, in which only part of the antioxidant compounds present in the leaves is extracted through the boiling water. Polyphenols analysis of the same samples reflects the ARP values previously showed (Figure 3).
Figure 1: Comparison of ARP values of Moringa and other two common South African beverages.
Figure 2: ARP values of leaf powder and a tea preparation from Moringa o.
Figure 3: Polyphenols concentration in the examined beverage preparations and the difference in Moringa o. leaf powder and tea.
Condensed tannins analysis
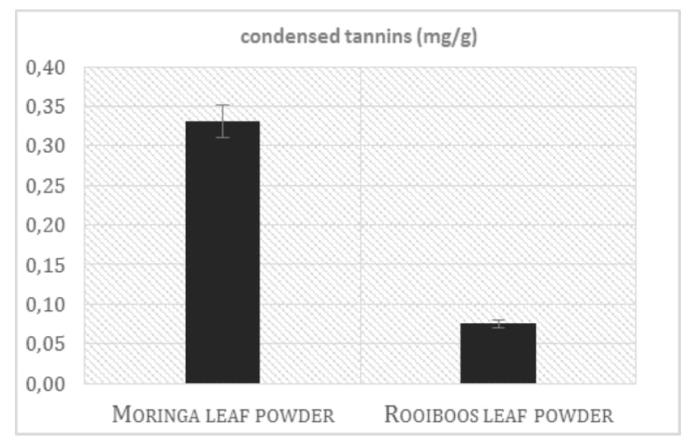
Moringa leaf powder was analysed for the condensed tannins content and compared with the same Rooibos material. As shown in the Figure 4, Moringa presents a concentration of tannins around 4 times higher respect to that found in Rooibos. These data about the tannins content can be considered in a twofold manner: condensed tannins represent one the major groups of antioxidant molecules found in the food and beverages, possessing multifunctional properties beneficial to human health, for instance it has been reported a reduction of the adverse effects of diabetes[36], but on the other side, they seem to be involved in the inhibition of the metabolism of digested and absorbed nutrients, particularly proteins[37]. Therefore, the nutritional property of the tannins present in Moringa extracts depends on type of consumed diet. Moreover, It is interesting to compare these results with those presented in literature about the content of tannins in the coffee, which contains, in fact, different concentrations of these polyphenolic compounds, according to the different varieties and roasting temperatures; as shown in the publication of Hecîmović et al., 2011[38], the pro-anthocyanidins content in the coffee is between 0.01 mg/g in the green cioccolato variety and 0.9 mg/g in Cherry variety, roasted at medium temperature.
Figure 4: Condensed tannins in Moringa and Rooibos leaf powder.
Free phosphate and phytic acid analysis
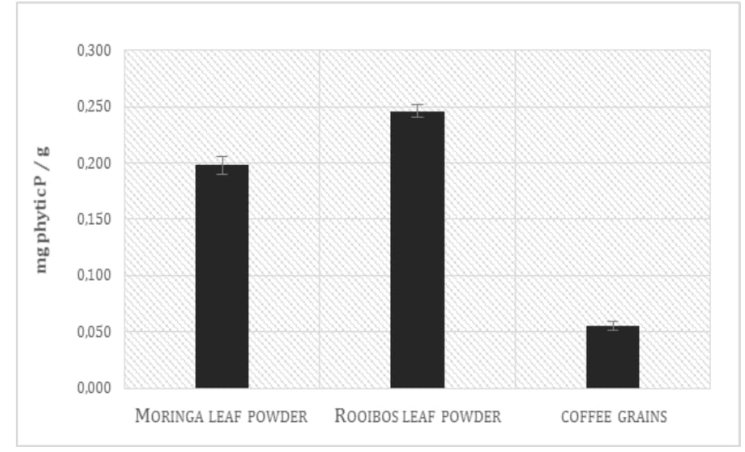
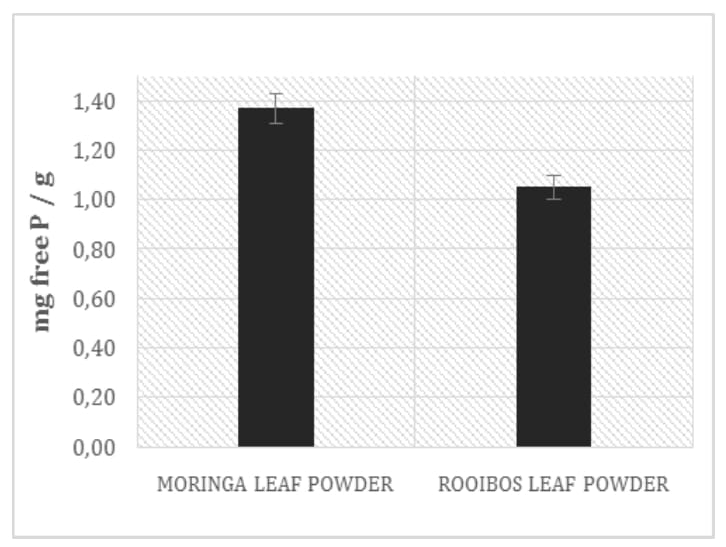
The phytic acid phosphate (PAP) content in Moringa leaf powder was determined and compared with that one found in Rooibos leaf powder and in coffee grains. Results, in the Figure 5, show a concentration of phytic acid in Rooibos tea leaves around 20% higher than in Moringa leaf powder (*p = 0.01) and around 80 higher than in coffee grains. Consequently, the free phosphate content observed in Moringa leaf powder is 24 higher than in Rooibos (***p = 0,00016) Figure 6. The finding of this study strengthen the argument about the higher nutritional quality of Moringa over Rooibos; Moringa oleifera extracts present a relevant amount of free phosphate and a lower content of phytic acid respect to those observed in Aspalathus linearis, considered, up to now, one of the most healthy beverage commonly used in South Africa and in Africa in general. Phytic acid, by virtue of its ability to chelate iron, is a potent inhibitor of the iron-driven formation of ROS, reactive oxygen species[39] and moreover it possess an important anticancirogenic activity; nevertheless it must be considered, in countries with a poor and monotonic diet,
Figure 5: Phytic acid content in M. oleifera, A.linearis and coffee grains.
Figure 6: Free phosphate content in Moringa and Rooibos leaf powder.
Ascorbic acid and flavonoids analysis:
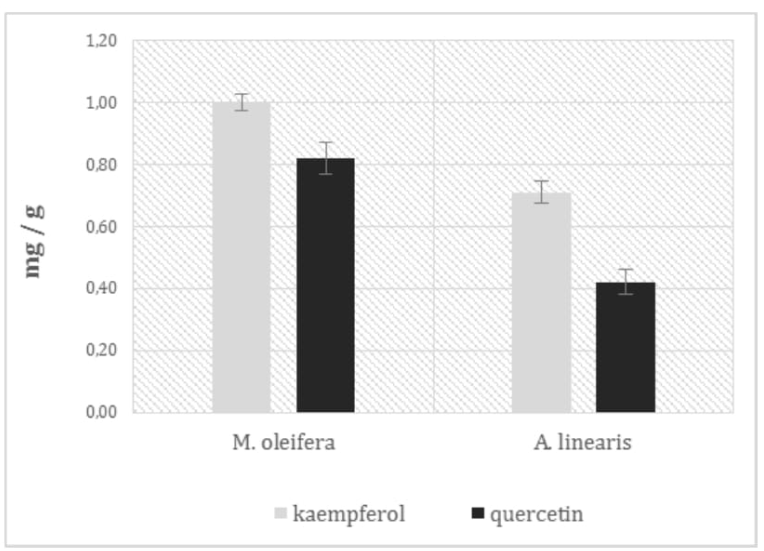
Moringa oleifera plant is well known to be rich in vitamin C; it possess more than 5 times the amount present in a typical orange juice beverage (data available at the following link: http://www.fao.org/docrep/x2650t/x2650t03.htm). Subsequently, a comparison test between the ascorbic acid content in Moringa and Rooibos leaf powder was then performed in this study. Obtained results showed a vitamin C concentration in Moringa 66.6 higher than in Rooibos (0.70 ± 0.04 mg/g and 0.42 ± 0.01 mg/g, respectively) (**p = 0,00037). These results about Moringa are slightly lower than those found in literature[40,41], but it is necessary to consider that the content of this vitamin can vary according to the examined Moringa genotype. Data regarding HPLC analysis of flavonoids from Moringa leaf powder were compared with results shown in literature for Rooibos. In the Figure 7, the two most important and abundant flavonoids, kaempferol and quercetin, were measured in Moringa leaf powder and compared with the values obtained in Rooibos leaf powder from Bramati[42]. Moringa plant presents an amount of the two flavonoids higher than in Rooibos (around 30 ) and, in both plants, kaemferol concentration is higher respect to the quercetin, around 20 higher in Moringa and 40 higher in Rooibos. These results about the flavonoids content in Moringa are compatible with those reported in literature; a recent publication shows how the different extraction methods affect the total flavonoids concentration from Moringa leaf dry powder, contributing to the variability of results[43].
Figure 7: Content of the main two flavonoids in Moringa and Rooibos leaves.
Trigonelline and ursolic acid analysis
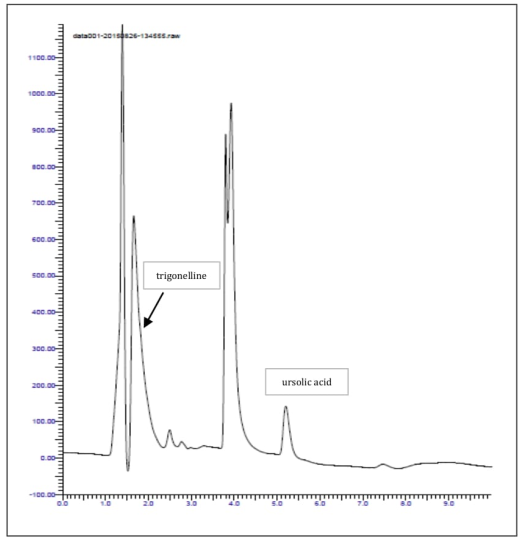
HPLC analysis (chromatogram illustrated in Figure 8) showed a relevant amount of these two compounds, especially of trigonelline, which reached the concentration of 2.83 mg/g. In the literature, the presence of trigonelline was not previously recorded in M. oleifera, except for a paper published from Mathur[44] in which trigonelline content was measured in various parts of the plant and in vitro cell cultures; in the leaves, the concentration of this alkaloid was around 2.6 mg/g, perfectly in line with results presented in this work.
Figure 8: HPLC analysis of ursolic acid and trigonelline in M. oleifera.
The measured value for ursolic acid content in Moringa leaf powder was 1.8 mg/g. This triterpenoid molecule is generally present in apple peel but also in several Lamiacee species; the results from this study is then one the first evidence of the presence of ursolic acid in a Moringaceae plants. In another published work the content of oleanolic acid and ursolic acid in Moringa, but using the seeds as experimental material, was analysed[45].
Antimicrobial activity
Moringa extract showed a significative antimicrobial and antifungal activity; the MIC values are reported in table 1.
Table 1:
| MIC values (mg/ml) | ||
|---|---|---|
| Mor. dry leaves extract | Mor. fresh leaves extract | |
| C. neoformans | 3.13 | 1.55 |
| C. albicans | 12.5 | 1.60 |
| E. fecalis | 12.5 | 6.25 |
| K. pneumoniae | 12.5 | 3.13 |
| S. aureus | 3.13 | 12.5 |
| P. aeroginosa | 3.13 | 50.0 |
In literature several studies about the antimicrobial action of the Moringa leaf extracts were published using different system of evaluation and different microbial strains[46,47,48]. The data from these studies showed a strong antimicrobial activity of Moringa, suggesting that these extracts can be used to discover antibacterial agent for developing new pharmaceuticals against human pathogens[49,50].
Conclusion
Moringa dry leaf extract, used in a commercial product in South Africa, was analysed for nutritional and healthy properties and some of these results were compared with those obtained from Rooibos and coffee. With the exception of the coffee, which present a much higher antioxidant activity respect to the other two plants, Moringa and Rooibos contain more or less the same concentration of antioxidant compounds. Moringa, instead, was found more rich in tannins, vitamin C and flavonoids quercetin and kaempferol respect to Rooibos.
The phytic acid content in Moringa resulted lower[28] than in Rooibos tea, with a consequent higher level of free phosphate. In this work, moreover, the concentration of trigonelline and ursolic acid in Moringa was detected setting up a HPLC method which allowed to determinate the presence of the two compounds in the same chromatographic run. These results showed for the first time a relevant amount of ursolic acid and confirmed the few data present in literature about the accumulation of trigonelline in the leaves. Finally, data regarding antimicrobial activity confirmed an important action of Moringa extracts against different strains of pathogens, bacteria and fungi. The overall results in this study highlight the potentials of Moringa plant extracts to mitigate the adverse effects of some diet and to contrast lifestyle related diseases.
Authors’ Contributions:
E.D. and B.D. conceived and designed the experiments; L.M. and
S.M. performed the experiments; E.D. and D.B., M.V., M.D. analyzed the data; E.D. wrote the paper. A.A.E. coordinated the project.
References
- 1. Allred, K,F., .Yackley, K, M., Vanamala, J.et al., Trigonelline is a novel phytoestrogen in coffee beans. (2009) J Nutr 139(10): 1833-1838.
- 2. Asghari, G., Palizban, A., Bakhshaei. B. Quantitative analysis of the nutritional components in leaves and seeds of the Persian Moringa peregrine (Forssk) Fiori. (2015) Pharmacognocy Res 7(3): 242-248.
- 3. Ampofo-Yeboah, A., Lambrechts, H., Brink, D., et al.,. Analysis of Oleanolic Acid and Ursolic Acid, Potential Antifertility Agents in Moringa (Moringa oleifera) Seed. (2013) J Agri Sci Tech 3: 989-999.
- 4. Bramati, L., Minoggio, M., Gardana,C., etal., Quantitative Characterization of Flavonoid Compounds in Rooibos Tea (Aspalathus linearis) by LC−UV/DAD. (2002) J Agric Food Chem 50(20): 5513−5519.
- 5. Brand-Williams, W., Cuvelier, M.E., Berset. C., Use of a free radical method to evaluate antioxidant activity. (1995) Food Sci Tech 28: 25–30.
- 6. Booth, F.E.M., Wickens. G.E.. Non-timber uses of selected arid zone trees and shrubs in Africa. (1988) In: FAO Conservation Guide. Food Agri Organi 176.
- 7. Butler, L.G. Antinutritional effects of condensed and hydrolyzable tannins. (1992) Basic Life Sci. 59:693-698.
- 8. Byun, J.O. and J.S. Han, 2004. A study perception and actual status of utilization for green tea. Korean Journal of Food Culture18: 184–192.
- 9. Caceres, A., Cabrera, O., Morales, O., Pharmacological properties of Moringa oleifera: Preliminary screening for antimicrobial activity. (1991) J Etnopharmacol 33(3): 213-216.
- 10. Chen, P.S., Toribara, T.Y., Warner, H. Micro-determination of phosphorus. (1956) Anal Chem 28(11): 1756–1758.
- 11. Doria, E., Galleschi, L., Calucci, L., etal.,. Phytic acid prevents oxidative stress in seeds: evidence from a maize (Zea mays L.) low phytic acid mutant. (2009) J Exp Botany 60(3): 967-978.
- 12. Doria, E., Campion, B., Sparvoli, F., Anti-nutrient components and metabolites with health implications in seeds of 10 common bean (Phaseolus vulgaris L. and Phaseolus lunatus L.) landraces cultivated in southern Italy. (2012) J Food Composition Anal 26(1–2): 72–80.
- 13. Eloff, J.N. Which extract should be used for the screening and isolation of antimicrobial components from plants. (1998) J Ethnopharmacol 60(1): 1-8.
- 14. Escarpa, A., Gonzales. M.C. High-performance liquid chromatography with diode-array detection for the determination of phenolic compounds in peel and pulp from different apple varieties. (1998) J Chromatog 823(1-2): 331–337.
- 15. Fahey, J.W. Moringa oleifera: A Review of the Medical Evidence for Its Nutritional, Therapeutic, and Prophylactic Properties. Part 1. (2005) Trees for life.
- 16. Gudej, J. Tomczyk, M. Determination of Flavonoids, Tannins and Ellagic Acid in Leaves from Rubus L. Species. (2004) Arch Pharm Res 27 (11): 1114-1119.
- 17. Hamilton-Miller, J.M. Antimicrobial properties of tea (Camellia sinensis. L). (1995) Antimicrob Agents Chemother 39 (11): 2375 – 2377.
- 18. Havsteen, B.H. The biochemistry and medical significance of the flavonoids. (2002) Pharmacol Ther 96(2-3):67-202.
- 19. Hecimovic´, I., Belšcak-Cvitanovic´, A., Horzic´ D., et al.,. Comparative study of polyphenols and caffeine in different coffee varieties affected by the degree of roasting. (2011) Food Chem 129(3): 991–1000.
- 20. Jung, D.H.. Components and Effects of Tea (In Korean), Hongikjae, Seoul. (2004) 28 – 43.
- 21. Khokhar, S., Magnusdottir. S.G. Total phenol, catechin, and caffeine contents of teas commonly consumed in the United Kingdom. (2002) J Agri Food Chem 50: 565 – 570.
- 22. Kumari, M. S. Jain. 2012. Tannins: An Antinutrient with Positive Effect to Manage Diabetes. Research Journal of Recent Sciences1 (12): 70-73.
- 23. Kunkel, S.D., Elmore, C.J., Adams. C.M.,. Ursolic acid increases skeletal muscle and brown fat and decreases diet-induced obesity, glucose intolerance and fatty liver disease. (2012) Plos One 7(6):e39332.
- 24. Marongiu, B.,S. Porcedda, A., Piras, A., Antioxidant activity of supercritical extract of Melissa officinalis. subsp. officinalis and Melissa officinalis subsp. Inodora (2004) Phytotherapy Research18 (10): 789 – 792.
- 25. Masoko, P. Eloff. J.N.Screening of 24 South African Combretum and 6 Terminalia (Combretaceae) species for antioxidant activities. (2007) Afr J Tradit Complement Altern Med 4(2): 231-239.
- 26. Mathur, M. Kamal. R.Studies on trigonelline from Moringa oleifera and its in vitro regulation by feeding precursor in cell cultures. (2012) J Pharmacognosy 22 (5): 994-1001.
- 27. Medoua, G. M., Egal, A. A., Oldewage-Theron. W. H.. Nutritional value and antioxidant capacity of lunch meals consumed by elderly people of Sharpeville. (2009) Food Chemistry 115(1): 260-264.
- 28. Middleton. E, Jr., Kandaswami, C., Theoharides. T.C.. The effects of plant flavonoids on mammalian cells: implications for inflammation, heart disease, and cancer. (2002) Pharmacol Rev 52(4): 673–751.
- 29. Morris, J.K., Mutton, D., Alberman. E.. Rates of Down syndrome at the upper extreme of maternal age: considerations and recommendations in analysis. (2006) Prenat Diag 26(11): 1091.
- 30. Ncube, B., Ngunge, V.N., Finnie, J.F., et al., A comparative study of the antimicrobial and phytochemical properties between outdoor grown and micropropagated Tulbaghia violacea.Harv Plants.( 2011) J Ethnopharmacol 134(3):775-780.
- 31. Newton, K.A. 2007. Effect of spacing and harvest frequency on the growth, leaf quality and yield of Moringa oleifera as a leafy vegetable. MSc. Thesis, Faculty of Agriculture and Natural Resources, KNUST. 4 – 6.
- 32. Ojiako, F.O., Adikuru N. C., Emenyonu. C. A.,. Critical issues in Investment, Production and Marketing of Moringa oleifera as an Industrial Agricultural raw material in Nigeria. (2011) J Africans Online 10 (2).
- 33. Orwa, C., Kindt, R., Jamnadass, R, H.,et al., Trichiliaemetica. Agroforestree database: a tree reference and selection guide version 4.0. 2009.
- 34. Osuna-Garcia, J. A., Wall, M. M., Waddell. A.. Endogenous Levels of Tocopherols and Ascorbic Acid during Fruit Ripening of New Mexican-Type Chile (Capsicum annuum L.) Cultivars. (1998) J Agric Food Chem. 46(12): 5093−5096.
- 35. Owusu, E., Odamtten. G.T. Quality of Ghana herbal tea: microflora and control measures. (1999) J Ghana Sci Assoc 1(3): 84-99.
- 36. Perez-Jimenez, J., Arranz, S., Tabernero, M., Updated methodology to determine antioxidant capacity in plantfoods, oils and beverages: Extraction, measurement and expression of results. (2008) Food Res Intern 41: 274–285.
- 37. Pilu, R., Landoni, M., Cassani, E.,. The Maize lpa241 Mutation Causes a Remarkable Variability of Expression and Some Pleiotropic Effects. (2005) Theor Appl Genet 45:2096–2105.
- 38. Rahman, M. M., Mominul, M. I. Sheik., Sharmin, S. A. et al., Antibacterial Activity of Leaf Juice and Extracts of Moringa oleifera Lam. against Some Human Pathogenic Bacteria. (2009) J Nat Sci 8(2): 219-227.
- 39. Razis, A.F.A., Ibrahim, M.D., Kntayya. S.B.,. Health benefits of Moringa oleifera. (2014) Asian Pac J Cancer Prev 15(20): 8571-8576.
- 40. Roberfroid, M.B., Concepts in functional foods: the case of inulin and oligofructose. (1999) J Nutrition 129 (7): 1398 – 1401.
- 41. Saini, R.K., Shetty, M., Prakash, P., et al.,Effect of dehydration methods on retention of carotenoids, tocopherols, ascorbic acid and antioxidant activity in Moringa oleifera leaves and preparation of a RTE product. (2014) J Food Sci Technol 51(9):2176-2182.
- 42. Singh,Y., Prasad K., Moringa Oleifera Leaf as Functional Food Powder: Characterization and Uses. (2013) J Agri Food Sci Tech 4(4): 317-324.
- 43. Smolin, L.A., Grosvenor. M.B. Nutrition: Science and applications, John Wiley Sons, 2003.
- 44. Swain, T., Hillis. W.E., The phenolic constituents of Prunus domestica. The quantitative analysis of phenolic constituents. (1959) J Sci Food Agr 10: 63–68.
- 45. Taralkar, S.V., Chattopadhyay. S.. A HPLC for determination of ursolic acid and betulinic acid from their methanolic extracts of Vitex negundo Linn. (2012) Anal\Bioanal Tech 3:3.
- 46. Thaiponga, K., Unaroj, B., Kevin, C., et al., Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. (2006) J Food Comp Anal 19(2006): 669–675.
- 47. Verdcourt, B., A synopsis of the Moringaceae. (1985) Kew Bulletin 40: 1-23.
- 48. Vongsaka, B., Sithisarna, P., Mangmoolb, S.,. Maximizing total phenolics, total flavonoids contents and antioxidant activity of Moringa oleifera leaf extract by the appropriate extraction method.(2013) Indus Crops Pro 44: 566– 571.
- 49. Waterman, P.G. S. Mole. 1994. Method in Ecology. Analysis of Phenolic Plant Metabolites. Blackwell Sci. Publ., London: 66–103.
- 50. Zhou, J., Chan, L., Zhou. S., Trigonelline: a plant alkaloid with therapeutic potential for diabetes and central nervous system disease. (2012) Curr Med Chem 19(21): 3523-3531.