An Overview of Venous Thrombo-Embolism Prophylaxis in Ambulatory Cancer Patients
Antoine Finianos1#, Firas Kreidieh1#, Ali Taher2*
Affiliation
1Department of Internal Medicine, American University of Beirut Medical Center, Beirut – Lebanon
2Naef K. Basile Cancer Institute, American University of Beirut Medical Center, Beirut – Lebanon
#Authors contributed equally
Corresponding Author
Ali Taher, Director, Naef K. Basile Cancer Institute, American University of Beirut Medical Center, Beirut – Lebanon; E-mail: fk30@aub.edu.lb
Citation
Taher, A., et al. An Overview of Venous Thrombo-Embolism Prophylaxisin Ambulatory Cancer Patients. (2020) Int J Cancer Oncol 7(1): 19-27.
Copy rights
© 2020 Taher, A. This is an Open access article distributed under the terms of Creative Commons Attribution 4.0
Keywords
Venous thrombo-embolism; Thromboprophylaxis; Ambulatory cancer
Abstract
Introduction: Patients with cancer are at higher risk of developing venous thrombo-embolism (VTE) with prevalence ranging between 15 and 20%(1-3).While bedridden and/or hospitalized cancer patients usually have standard prophylactic and therapeutic guidelines, ambulatory patients, who undergo chemotherapy, hormonal therapy, immunotherapy, or radiotherapy, do not have standard indications for prevention. In this review article, we aim at providing an overview of venous thrombo-embolic complications in ambulatory cancer patients, unveiling the available VTE risk scores and options for prophylaxis.
Body: There are several risk stratification scores that were studied for ambulatory cancer patients. The modified Khorana score is currently recommended by the American Society of Clinical Oncology; however, this score was shown in some studies to perform poorly in some cancers like lung cancer (30). Patients with a solid tumor and risk score of ≥2 have an estimated risk of symptomatic thrombosis of 9.6% during the first 6 months of chemotherapy. The role of LMWH, DOACs, and vitamin K antagonists for thromboprophylaxis in ambulatory cancer patients has been studied with varying results. Both, the European Society of Medical Oncology and the American Society of Medical Oncology do not recommend the routine use of thromboprophylaxis for ambulatory cancer patients. The CASSINI and AVERT trials have shown that thromboprophylaxis with DOACs is relatively effective and safe in the population of cancer patients with Khorana score ≥2. However, it may be safer to use them in patients with non-GI or GU cancers as it is consistently proven that DOACs are associated with increased risk of bleeding in these tumor types. Both trials had different tumor types distribution and methodology thus affecting outcomes though subgroup analysis of pancreatic cancer patients in the CASSINI trial revealed no increased bleeding with rivaroxaban.
Conclusion: Risk assessment using predictive scores to identify high risk ambulatory cancer patients and implementing thereafter a patient-centered approach is recommended. Discussion with each patient the risks versus the benefits of thromboprophylaxis should be the basis of initiation of thromboprophylaxis. Conducting randomizing trials assessing DOACs in individual cancer subtypes may be the best method to provide definitive evidence about their efficacy and safety. .
Introduction
Patients with cancer are at higher risk of developing Venous Thrombo-Embolism (VTE) with prevalence ranging between 15 and 20%[1-3]. The first observation for thrombosis in cancer patients was made in 1865 by Armand Trousseau who described an association between the activation of blood coagulation cascade and tumor growth. His observation related primary or idiopathic VTE to the possible diagnosis of an underlying occult malignancy[2]. Since then, VTE has represented a well-recognized complication of cancer and a possible initial presentation of underlying malignancy[4]. Thrombosis carries a high rate of morbidity and mortality in cancer patients and represents the second cause of death in these patients[2].
Malignancy is a known thrombophilic condition. The absolute risk of VTE generally depends on the stage or extent of disease and the cancer type with higher rates in pancreatic, gastric, kidney, and brain tumors[1]. This risk association can also be attributed to multiple factors, including increased platelet activation, increased cytokine production, and over-expression of tissue factor in cancer patients. The presence of central venous catheters, cancer-related surgeries, chemotherapy, and hormonal therapy can increase the risk in this patient population as well[4,5]. For example, surgery for cancer patients can be associated with at least double the risk of postoperative deep vein thrombosis (DVT) and at least three times the risk of fatal pulmonary embolism when compared to patients without cancer[1].
The substantial variation in thrombosis risk of cancer patients due to the multitude of contributing factors on one hand, and the morbidity and mortality that VTE can carry on the other, entail the need for appropriate risk stratification, prevention, and management in this patient population. While bedridden and/or hospitalized cancer patients usually have standard prophylactic and therapeutic guidelines, ambulatory patients, who undergo chemotherapy, hormonal therapy, immunotherapy, or radiotherapy, do not have standard indications for prevention.
Objectives
In this review article, we aim at providing an overview of venous thrombo-embolic complications in ambulatory cancer patients, unveiling the available VTE risk scores and options for prophylaxis.
Methodology
We conducted an electronic literature search of PubMed (Ovid), MEDLINE (Ovid), and Embase (Ovid) databases, in addition to the Cochrane Database of Systematic Reviews 2017, Issue 9. The following Medical Subject Headings (MeSH) terms were used: “low-molecular-weight heparin”, “unfractionated heparin”, “direct oral anticoagulants”, “thromboprophylaxis”, “ambulatory cancer”, “venous thromboembolism”, “deep vein thrombosis”, “pulmonary embolism”, and “Khorana risk score”. We included randomized controlled trials that enrolled ambulatory cancer patients and assessed the effect of low‐molecular weight heparin (LMWH), unfractionated heparin (UFH), and Direct Oral Anticoagulants (DOACs) on mortality, deep venous thrombosis (DVT), pulmonary embolism (PE), bleeding outcomes, and thrombocytopenia. Clinical guidelines published by the American Society of Clinical Oncology and the European Society of Medical Oncology were also included.
Risk Scores for VTE in ambulatory cancer patients
While prophylaxis against VTE reduces its risk, current consensus guidelines generally recommend against thromboprophylaxis in ambulatory cancer patients. These guidelines were based on the risk of bleeding, the high number needed to treat to prevent an event of thrombosis which can reach 50 patients, and the added burden of daily heparin injection[6,7]. As a result, risk stratification would help guide clinicians identify patients with high risk for VTE who deserve thromboprophylaxis.
To date, the most widely used tool is the Khorana Risk Score, which was initially introduced in 2008 by Alek Khorana and colleagues[8]. Table 1 shows the modified risk stratification model for cancer-associated VTE in the ambulatory setting.
Table 1: Modified risk stratification model for cancer-associated VTE in the ambulatory setting - Khorana Thromboembolic Risk Score(6)
|
Patient characteristic |
Score |
|
Cancer |
|
|
• Very high risk: stomach, pancreas • High risk (lung, lymphoma, gynecologic, bladder, testicular, renal tumors) |
2 1 |
|
Pre-chemotherapy platelet count >350,000/Ìl |
1 |
|
Hemoglobin level < 10 g/dL or use of red cell growth factors |
|
|
Pre-chemotherapy leukocyte count > 11,000/Ìl |
1 |
|
Body mass index >35 kg/m2 |
1 |
|
Calculate total score, adding points for each criterion in the model |
|
In order to assess the utility of this risk score as a predictor for VTE, a large meta-analysis by Mulder et al evaluated 34000 ambulatory cancer patients from over 50 studies between January 2008 and June 2018. The risk of VTE in patients who had elevated Khorana score was 11.0% which was significantly greater than the risks of those with intermediate and low scores, 6.6 and 5.1%, respectively. Despite this, of all patients who developed VTE within the first six months, only around one-quarter (23.4%) had a high Khorana risk score[6,9]. Moreover, the Khorana score was found in certain cancers not to be a valid predictive model for VTE, as in lung cancer and GBM. For example, a retrospective study assessing risk factors for VTE during adjuvant therapy for GBM, the Khorana score was an invalid predictive model in these patients with poor specificity[10].
To improve on the utility of risk calculations, some authors have proposed other scores, namely the Vienna Cancer and Thrombosis Study (CATS), PROphylaxis of ThromboEmbolism during ChemoTherapy (PROTECHT), and ChariteOnkologie (CONKO) scores, that include biomarker measurements, the type of chemotherapy, or replacement of the BMI with the performance status[7]. The Vienna CATS score includes, in addition to the parameters of Khorana, a D-dimer level that is greater than 1.44 μg/L (1 point) and a Soluble P-Selectin level that is greater than 53.1 ng/L (1 point). The PROTECHT score includes, in addition to the parameters of Khorana, Gemcitabine chemotherapy (1 point) and Platinum-based chemotherapy (1 point).
CONKON score replaces the BMI parameter with World Health Organization (WHO) performance status that is greater than or equal to 2 (1 point) [7]. In a multi-national prospective cohort study, Van Es et al evaluated and compared the risk scores in 876 patients from whom 260 patients were enrolled prior to initiation of chemotherapy. Of these patients, 20 (7.7%) developed VTE during a six months follow-up period. At a threshold of 3 points, the scores classified patients as high risk in 13 to 34% of patients depending on the score.
The scores had a poor discriminatory performance although the Vienna CATS and PROTECHT scores were better able to discriminate high and low risk patients. In fact, high risk patients had a significantly increased risk of VTE when using the Vienna CATS score (HR of 1.7; 95% CI:1.0-3.1) and PROTECHT score (HR of 2.1; 95% CI: 1.2-3.6). The results of this meta-analysis do not support the use of these scores to select patients for thromboprophylaxis[7].
In an attempt to improve these scores, Pabinger et al analyzed data from the Vienna Cancer and Thrombosis Study (CATS) to select prognostic parameters for inclusion into their model. This model was validated by using the prospective Multinational Cohort Study to Identify Cancer Patients at High Risk of Venous Thromboembolism (MICA) cohort. They developed a simple clinical prediction model consisting of one clinical factor, tumor-site category, and one biomarker, D-dimer level. This externally validated model predicted the risk of VTE in ambulatory patients with solid malignancies. It is a promising model that may be used as a predictor for VTE to select who warrants thromboprophylaxis. The predicted cumulative six-month risk of VTE in Vienna CATS was set at a 10% cutoff. The model had a sensitivity of 33%, specificity of 84%, a positive predictive value of 12%, and a negative predictive value of 95%[11]. This is a novel model for clinical prediction of VTE in ambulatory cancer patients.
Patients with a solid tumor and risk score of more than or equal to 2 have an estimated risk of symptomatic thrombosis of 9.6% during the first 6 months of chemotherapy[12]. In addition, the incidence of VTE in ambulatory cancer patients receiving chemotherapy increases with the number of cycles of chemotherapy received with a cumulative rate of 2.2% (95% CI 1.7-2.8) according to a study by Khorana et al[13]. The current American Society of Clinical Oncology practice guidelines recommend the Khorana score as the only validated risk assessment tool for the prediction of cancer-associated VTE in ambulatory patients[14]. Figure 1 shows a schematic diagram that compares the risk of VTE in ambulatory cancer patients during the course of their disease.
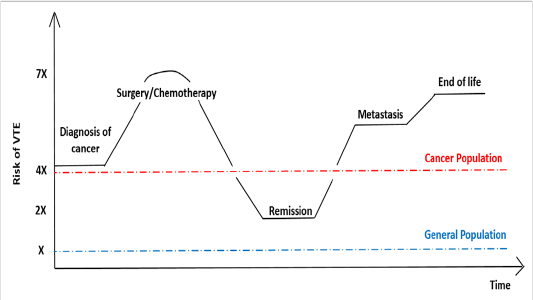
Figure 1: Schematic diagramcomparing the of risk of VTE in ambulatory cancer patients during the course of their disease[15-17]
Thromboprophylaxis in ambulatory cancer patients
While thromboprophylaxis is most often recommended in hospitalized surgical and non-surgical cancer patients, there is lack of clear data with low grade recommendation against routine use of anticoagulants in ambulatory cancer patients[9]. These patients usually receive non-surgical management, including radiation therapy and chemotherapy in an outpatient or short-stay hospital setting. Their management includes the use of long-term central venous catheters through which chemotherapy is administered. As a result, there is an increased risk of DVT involving the internal jugular and the subclavian veins, which can reach up to 20% without symptoms and 5% with clinical manifestations of thrombosis[9]. While studies have shown a significant drop in the risk of asymptomatic catheter-related DVT with Low Molecular Weight Heparin (LMWH) and Vitamin K antagonists, they did not support their use to prevent symptomatic catheter-related DVT. Hence, international guidelines recommend against their use for prevention of catheter-associated DVT, for example[15-18].
The role of thromboprophylaxis in ambulatory cancer patients receiving chemotherapy as outpatients has been studied with a variation in results. A meta-analysis by Akl et al included 9575 patients to evaluate the efficacy and safety of parenteral anticoagulants in ambulatory cancer patients. Heparin therapy reduces the risk of symptomatic VTE with a relative risk RR of 0.56 (95% CI 0.47 to 0.68) and risk difference RD of 30 fewer per 1000 (95% CI 36 fewer to 22 fewer) with high certainty of evidence. However, this came at the expense of increase in the risks of major bleeding with RR of 1.30 (95% 0.94 to 1.79) and RD of 4 more per 1000 (95% CI 1 fewer to 11 more) with moderate certainty of evidence and of minor bleeding with RR of 1.70 (95% 1.13 to 2.55) and RD of 17 more per 1000 (95% CI 3 more to 37 more) with high certainty of evidence[19].
The role of Low-Molecular-Weight-Heparin (LMWH) varied in different studies with the type of malignancy. While it showed no benefit for breast and lung cancer patients in a study by Haas et al, the PROTECHT study showed that Nadroparin resulted in 50% risk reduction in pancreatic, breast, ovarian, gastrointestinal, and head and neck cancers[9,20]. In addition, a novel LMWH, semuloparin, resulted in 65% risk reduction for patients with pancreatic, gastric, colorectal, bladder, and ovarian cancer[9]. The incidence of clinically relevant bleeding was 2.8% and 2.0% in the semuloparin and placebo groups, respectively[21]. These two large trials showed some benefit for VTE prophylaxis for nadroparin and semuloparin, respectively, with low event rates.
To study if parenteral thromboprophylaxis may have a greater benefit for patients with high risk for VTE, Khorana et al investigated the role of dalteparin in patients with Khorana score greater than or equal to 3. Patients were randomized to 5000 units subcutaneously daily or to observation for 12 weeks. VTE occurred in 12% of patients who received dalteparin and in 21% of patients in the observation group with HR of 0.69 (95% CI 0.23-1.89), but this did not reach statistical significance. There was, however, a statistically significant increased risk for clinically relevant bleeding (HR 7, 95% CI 1.2-131.6)[22]. What is interesting in the aforementioned study, is that the number needed to treat decreased to around 12 to 15 when higher risk patients were selected. Despite these studies, international guidelines do not recommend thromboprophylaxis for ambulatory cancer patients. Table 2 summarizes findings regarding LMWH thromboprophylaxis in different clinical settings, and table 3 summarizes clinical trials that investigated the role of LMWH for thromboprophylaxis in ambulatory cancer patients.
Table 2: Low-Molecular-Weight-Heparin Thromboprophylaxis in different clinical settings
|
Clinical Setting |
Change in Incidence of VTE |
Number Needed to Treat (NNT) |
|
Postoperative [23] |
5.6% to 2.6% |
40 |
|
Hospitalized [9] |
5.0% to 2.8% |
45 |
|
Outpatient on treatment[24] |
3.9% to 2.0% |
50-60 |
|
Outpatient on treatment with Khorana score ≥3[25] |
21% to 12% |
12-15 |
Table 3: Clinical Trials for Low-Molecular-Weight-Heparin Thromboprophylaxis
|
LMWH |
Clinical Trial |
Cancer Type |
Clinical Outcome |
Adverse Events |
|
Bemiparin |
Lecumberri 2013 |
Limited-stage small cell lung cancer |
· Median progression-free survival: 272 days with chemoradiotherapy alone and 410 days in the bemiparin group (HR 2.58; 95% CI, 1.15-5.80; p = 0.022) · Median overall survival: 345 days with chemoradiotherapy alone and 1133 days in the bemiparin group (HR 2.96; 95% CI, 1.22-7.21; p = 0.017) · Rate of tumor response was similar in both study arms |
· No significant between-group difference in the rates of major bleeding · Toxicity related with the experimental treatment was minimal |
|
Certoparin |
TOPIC-1 |
Disseminated metastatic breast carcinoma |
· VTE incidence: not different between treatment groups (7 of 174 (4%) treated with Certoparin compared to 7 of 177 (4%) receiving placebo (OR 1.02; 95% CI 0.30-3.48) |
· Mortality not different between groups |
|
|
TOPIC-2 |
Stage III or IV non–small-cell lung carcinoma |
· VTE incidence: 12 of 268 (4.5%) versus 22 of 264 (8.3%) for Certoparin and placebo groups, respectively (OR 0.52; CI 0.23-1.12) |
· Mortality not different between groups · No significant increase in clinical bleeding |
|
Dalteparin |
Altinbas 2004 |
Small cell lung cancer |
· Median overall survival was 8.0 months with chemotherapy alone and 13.0 months with Dalteparin (p=0.01). · Similar improvement in survival with Dalteparin treatment occurred in patients with both limited and extensive disease stages |
· Risk of death in the Dalteparin group relative to that in the chemotherapy alone group was 0.56 (95% CI 0.30, 0.86) (p=0.012) · Toxicity from the experimental treatment was minimal · No treatment-related deaths |
|
Kakkar 2004 |
Stage III or IV malignant disease of the breast, lung, gastrointestinal tract, pancreas, liver, genitourinary tract, ovary, or uterus |
· 1, 2, and 3 years survival for Dalteparin group were 46%, 27%, and 21%, respectively, compared with 41%, 18%, and 12%, respectively, for patients receiving placebo (P=0.19) ·VTE incidence: 2.4% and 3.3% for dalteparin and placebo, respectively |
· Bleeding rates: 4.7% (major 0.5% and minor 4.2%) and 2.7% (major 0% and minor 2.7%) for Dalteparin and placebo groups, respectively |
|
|
Sideras 2006 |
Advanced breast, prostate, lung, or colorectal cancer |
· Median survival time: 10.5 months (95% CI 7.6-12.2 months) and 7.3 months (95% confidence interval, 4.8-12.2 months) for the placebo and Dalteparin groups, respectively · Rate of severe or life-threatening VTE: 6% in Dalteparin arm and 7% in the control arm |
· Rate of severe or life-threatening bleeding: 3% in the Dalteparin arm and 7% in the control arm |
|
|
Perry 2010 (PRODIGE) |
Malignant glioma |
· VTE occurrence: Twenty-two patients developed VTE in the first 6 months; nine in the Dalteparin group and 13 in the placebo group (HR= 0.51, 95% CI: 0.19–1.4, P = 0.29) |
At 12 months, 5 (5.1%) major bleeds on Dalteparin and 1 (1.2%) on placebo occurred (HR = 4.2, 95% CI: 0.48–36, P = 0.22) · All major bleeds were intracranial and occurred while on study medication · 12-month mortality rates: 47.8% for Dalteparin and 45.4% for placebo (HR = 1.2, 95% CI: 0.73–2.0, p = 0.48) |
|
|
Macbeth 2016 (FRAGMATIC) |
Newly diagnosed lung cancer of any stage and histology |
· No evidence of difference in overall or metastasis-free survival between the two arms (HR, 1.01; 95% CI, 0.93 to 1.10; p = 0.814; and HR, 0.99; 95% CI, 0.91 to 1.08; p = .864, respectively) · Reduction in the risk of VTE from 9.7% to 5.5% (HR, 0.57; 95% CI, 0.42 to 0.79; p = 0.001) in the Dalteparin arm |
· No difference in major bleeding events · Evidence of an increase in the composite of major and clinically relevant non-major bleeding in the Dalteparin arm |
|
|
Bozas 2016 |
Advanced metastatic pancreatic cancer |
· VTE incidence: 13% and 7% for patients who did not receive Dalteparin and those who received it, respectively |
· Bleeding incidence: 11% for both arms · Majority of the bleeding events observed due to cancer related lesions (duodenal infiltration or varices) |
|
|
Enoxaparin |
Zwicker 2013 |
Pancreatic adenocarcinoma (locally advanced or metastatic), colorectal (stage IV), non-small cell lung cancer (stage III or IV), relapsed or stage IV ovarian, or surgically unresectable or metastatic gastric adenocarcinoma. |
· Cumulative incidence of VTE at 2 months in the higher tissue-factor bearing microparticles (TFMP) group randomized to enoxaparin (N=23) was 5.6% while the higher TFMP group observation arm (N=11) was 27.3% (Gray test P=0.06) · Cumulative incidence of VTE in the low TFMP was 7.2% (N=32) · Median survival for patients with higher levels of TFMP followed by observation was 11.8 months compared with 17.8 months on enoxaparin (P=0.58) |
· No major hemorrhages observed in the enoxaparin arm |
|
Pelzer 2015 |
Advanced pancreatic cancer |
· Overall cumulative incidence rates of symptomatic VTE: 15.1% (observation group) and 6.4% (enoxaparin; HR, 0.40; 95% CI, 0.19 to 0.83; P =0.01) · Progression-free survival (HR, 1.06; 95% CI, 0.84 to 1.32; P=0.64) and overall survival (HR, 1.01; 95% CI, 0.87 to 1.38; P=0.44) did not differ between groups |
· Numbers of major bleeding events: five of 152 patients in the observation arm and seven of 160 patients in the enoxaparin arm (HR, 1.4; 95% CI, 0.35 to 3.72; p=1.0). |
|
|
Nadroparin |
Agnelli 2009 |
Lung, gastrointestinal, pancreatic, breast, ovarian, or head and neck |
· VTE incidence: 15 (2.0%) of 769 patients treated with Nadroparin and 15 (3.9%) of 381 patients treated with placebo (single-sided p=0.02) |
· 5 (0.7%) of 769 patients in the nadroparin group and no patients in the placebo group had a major bleeding event (two-sided p=0·18) · Minor bleeding incidence: 7·4% (57 of 769) with nadroparin and 7·9% (30 of 381) with placebo · 121 (15·7%) serious adverse events (SAE) in the nadroparingoup and 67 (17·6%) serious adverse events in the placebo group ·Types of SAE: abdominal pain, asthenia, condition aggravated, dyspnea, fever, intestinal obstruction, Neutropenia |
|
Frederiek 2011 |
Stage IIIb Non–small-cell lung cancer, hormone-refractory prostate cancer, or locally advanced pancreatic cancer |
· Median survival: 13.1 months in the Nadroparin recipients compared with 11.9 months in the no-treatment arm (HR 0.94; 95% CI, 0.75 to 1.18, adjusted for cancer type · No difference in time to progression |
· Number of major bleeding comparable at 4.1% in the nadroparin arm and 3.5% in the control arm |
|
|
Semuloparin |
Agnelli 2012 (SAVE-ONCO) |
Metastatic or locally advanced cancer of the lung, pancreas, stomach, colon or rectum, bladder, or ovary |
· VTE incidence: 20 of 1608 patients (1.2%) receiving semuloparin and 55 of 1604 (3.4%) receiving placebo (hazard ratio, 0.36; 95% confidence interval [CI], 0.21 to 0.60; P<0.001) |
· Clinically relevant bleeding incidence: 2.8% and 2.0% in the semuloparin and placebo groups, respectively (hazard ratio, 1.40; 95% CI, 0.89 to 2.21) · Major bleeding incidence: 19 of 1589 patients (1.2%) receiving semuloparin and 18 of 1583 (1.1%) receiving placebo (hazard ratio, 1.05; 95% CI, 0.55 to 1.99) · Incidences of all other adverse events: similar in the two study groups |
|
Subcutaneous Heparin |
Lebeau 1994 |
Small cell lung cancer |
· Response rate: 37% versus 23% for the heparin group versus those who did not receive heparin (p= 0.004) · Median survival (317 days vs. 261 days, respectively, p = 0.01) |
· No important bleeding or thrombocytopenia related to heparin treatment |
Direct Oral Anticoagulants (DOACs) in thromboprophylaxis
With the modest decrease in VTE risk with parenteral anticoagulants, on one hand, and the risk of bleeding and inconvenience of injections, on the other, the need to search for alternative options for thromboprophylaxis ensues. As a result, multiple trials have been conducted to investigate the role of DOACs for thromboprophylaxis in ambulatory cancer patients. DOACs include direct factor Xa inhibitors, namely rivaroxaban, apixaban, edoxaban, and betrixaban, and direct thrombin inhibitors, namely argatroban, bivalirudin, and dabigatran (Figure 2).
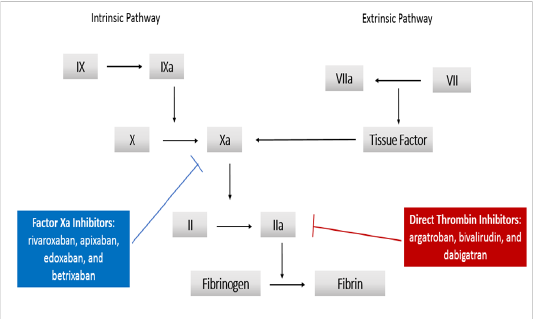
Figure 2: Direct Oral Anticoagulants (DOACs)
Safety and efficacy of rivaroxaban prophylaxis in ambulatory cancer patients were assessed by the CASSINI trial. This was a multicenter, multinational, randomized, double-blind, placebo-controlled phase IIIb superiority trial, that included patients at high risk for VTE (Khorana score ≥ 2), with an ECOG performance status of 0-2 and a negative baseline compression ultrasonography. The baseline characteristics of the rivaroxaban and placebo groups were well-balanced though more patients with a history of VTE were randomly assigned to the rivaroxaban arm.Of the 841 randomized patients, the primary end point occurred in 25 of 420 patients (6.0%) in the rivaroxaban group and in 37 of 421 (8.8%) in the placebo group, with a HR of 0.66; 95% CI, 0.40 to 1.09; p = 0.10, in the period up to day 180. In the prespecified intervention-period analysis that was from the day of first receipt of rivaroxaban to last dose plus two days, the primary end point occurred in 11 patients (2.6%) in the rivaroxaban group and in 27 (6.4%) in the placebo group, with a HR of 0.40; 95% CI, 0.20 to 0.80. Therefore, rivaroxaban significantly reduced VTE during the treatment period but not during full study period. Major bleeding occurred in 8 of 405 patients (2.0%) in the rivaroxaban group and in 4 of 404 (1.0%) in the placebo group (HR1.96; 95% CI, 0.59 to 6.49). Khorana score cutoff of more than or equal to 2 can be used to identify patients with high risk for VTE, and the study can serve as basis for recommendations for thromboprophylaxis in high risk ambulatory cancer patients[26].
Different outcomes were obtained in another trial, AVERT (Apixaban for the Prevention of Venous Thromboembolism in High-Risk Ambulatory Cancer Patients). This was a randomized, double-blind, placebo-controlled phase IIIb superiority trial. This study included ambulatory adult patients with newly diagnosed cancer or with progression of known cancer after complete or partial remission, and who were initiating a new course of chemotherapy with a minimum treatment intent of 3 months and are at high risk of VTE (Khorana score ≥ 2). Of the 563 patients who were included in the modified intention-to-treat analysis, VTE occurred in 12 of 288 patients (4.2%) in the apixaban group and in 28 of 275 patients (10.2%) in the placebo group, with a HR of 0.41; 95% CI, 0.26 to 0.65; P < 0.001. In the modified intention-to-treat analysis, major bleeding occurred in 10 patients (3.5%) in the apixaban group and in 5 patients (1.8%) in the placebo group (HR 2.00; 95% CI, 1.01 to 3.95; P = 0.046). Therefore, thromboprophylaxis with apixaban resulted in a significantly lower rate of VTE complications as compared to placebo. The rate of major bleeding was significantly higher with the apixaban group versus placebo in the modified intent-to-treat analysis, 3.5% versus 1.8%, with a HR of 2.00, but the rate was not significantly higher in the analysis of outcomes during the treatment period[27]. Table 4 summarizes clinical trials that investigated the role of DOACs for thromboprophylaxis in ambulatory cancer patients.
Table 4: Clinical Trials for DOACs Thromboprophylaxis
|
Clinical Trial |
DOAC |
Cancer Type |
Clinical Outcome |
Adverse Events |
|
Carrier 2019 (AVERT ) |
Apixaban |
Any solid malignancy |
· VTE incidence: 12 of 288 patients (4.2%) in the apixaban group and 28 of 275 patients (10.2%) in the placebo group, with a HR of 0.41; 95% CI, 0.26 to 0.65; P<0.001 |
· Major bleeding: 6 patients (2.1%) in the apixaban group and 3 patients (1.1%) in the placebo group (hazard ratio, 1.89; 95% CI, 0.39 to 9.24) during the treatment period |
|
Khorana 2019 (CASSINI) |
Rivaroxaban |
Any solid malignancy except primary brain tumor |
· VTE incidence: 25 of 420 patients (6.0%) in the rivaroxaban group and 37 of 421 (8.8%) in the placebo group (HR0.66; 95% CI, 0.40 to 1.09; p=0.10) in the period up to day 180 · In high-risk ambulatory patients with cancer, treatment with rivaroxaban did not result in a significantly lower incidence of VTE or death due to VTE in the 180-day trial period · In the prespecified intervention-period analysis, VTE incidence was 2.6% in the rivaroxaban group and in 6.4% in the placebo group, with a HR of 0.40; 95% CI, 0.20 to 0.80 |
· Major bleeding: 8 of 405 patients (2.0%) in the rivaroxaban group and 4 of 404 (1.0%) in the placebo group (HR1.96; 95% CI, 0.59 to 6.49) |
There were many differences between both trials that could explain the differences in outcomes. First, the higher rates of bleeding in GI and GU tracts with both DOACs is mainly explained by the fact that the most common primary cancer in the CASSINI trial was pancreatic cancer (32.6%) while in the AVERT trial, gynecologic cancers comprised the majority (25.8%). The rate of high GI/GU bleeding with DOACs in cancer patients is in conjunction with the findings of the Hokusai study assessing Edoxaban VTE treatment in cancer patients. Unlike the CASSINI trial, the AVERT trial used modified intention-to-treat analysis including participants who received at least one dose of the study medication. Moreover, the CASSINI trial, included patients with existing asymptomatic VTE as it required baseline ultrasound screening for its participants[26,27].
Upon assessing both trials together, the cumulative relative risk of VTE in the intention-to-treat analysis or during the treatment period, both DOACs were associated with improved outcome (RR 0.56[0.38-0.83] NNT 24, RR 0.29[0.16-0.53] NNT 21, respectively). Although the risk of bleeding is higher with both DOACs, there were no difference in mortality between the DOACs and placebo[28]. Even though DOACs are associated with increased bleeding especially in upper GI cancers as evident in the prophylaxis and VTE treatment studies, prespecified subgroup analysis of pancreatic cancer patients in the CASSINI trial revealed otherwise. Major bleeding occurred in 2 (1.5%) patients with pancreatic cancer on rivaroxaban versus 3 (2.3%) in the placebo arm during the intervention period. This further stresses the need for dedicated cancer site-specific randomized trials to assess the efficacy and side effects of DOACs in this population of patients[29].
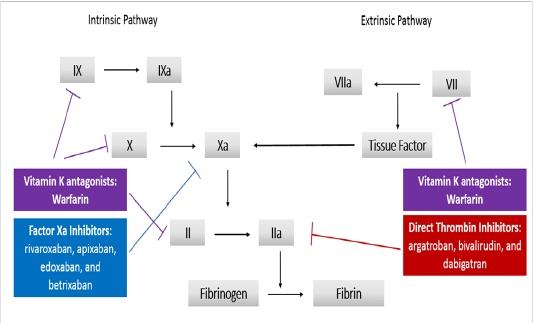
Figure 3: Mechanisms of Anticoagulants
The American Society of Oncology (ASCO) and the European Society of Medical Oncology (ESMO) recommend considering thromboprophylaxis for solid cancer patients receiving chemotherapy as outpatients and who have additional risk factors[9]. In this context, risk-adapted approach would be best and the use of the previously mentioned risk scores. According to a recently published update on ASCO clinical practice guidelines, there is strong recommendation not to offer routine pharmacologic prophylaxis to all ambulatory cancer patients. For high-risk ambulatory cancer patients, there is intermediate to high quality evidence for apixaban and rivaroxaban and intermediate quality evidence for LMWH consideration provided that there are no significant risk factors for bleeding or drug interaction[30]. Table 5 summarizes the recommendations by ASCO and ESMO.
Table 5: Summary of recommendations by ASCO and ESMO (Ref 9)
|
Patient Population |
ASCO |
ESMO |
|
Ambulatory Cancer Patients |
Not recommend routine thromboprophylaxis |
Not recommend routine thromboprophylaxis |
|
Ambulatory Cancer Patients with additional risk factors |
• Discussion with each patient the risks versus benefits of thromboprophylaxis • Patient-centered approach with each case dealt with differently |
• Risk assessment using predictive scores to identify high risk ambulatory cancer patients • Patient-centered approach with each case dealt with differently |
Conclusion
Patients with cancer are at higher risk of developing VTE with prevalence ranging between 15 and 20% of cancer patients and with a high rate of morbidity and mortality for ambulatory cancer patients[1-3]. The substantial variation in thrombosis risk of cancer patients due to the multitude of contributing factors on one hand, and the morbidity and mortality that DVT and VTE can carry on the other, entail the need for appropriate risk stratification, prevention, and management in this patient population. There are several risk stratification scores that were studied for ambulatory cancer patients. The modified Khorana score is currently recommended by the American Society of Clinical Oncology; however, this score was shown in some studies to perform poorly in some cancers like lung cancer[31]. Patients with a solid tumor and risk score of ≥ 2 have an estimated risk of symptomatic thrombosis of 9.6% during the first 6 months of chemotherapy. The role of LMWH, DOACs, and vitamin K antagonists for thromboprophylaxis in ambulatory cancer patients has been studied with varying results. Both, the European Society of Medical Oncology and the American Society of Medical Oncology do not recommend the routine use of thromboprophylaxis for ambulatory cancer patients. Risk assessment using predictive scores to identify high risk ambulatory cancer patients and implementing thereafter a patient-centered approach is recommended. Discussion with each patient the risks versus the benefits of thromboprophylaxis should be the basis of initiation of thromboprophylaxis. The CASSINI and AVERT trials have shown that thromboprophylaxis with DOACs is relatively effective and safe in the population of cancer patients with Khorana score ≥ 2. However, it may be safer to use them in patients with non-GI or GU cancers as it is consistently proven that DOACs are associated with increased risk of bleeding in these tumor types. Both trials had different tumor types distribution and methodology thus affecting outcomes. Probably, conducting randomizing trials assessing DOACs in individual cancer subtypes may be the best method to provide definitive evidence about their efficacy and safety.
Ethical Statement: Our study did not require an ethical board approval because it did not contain human or animal trials
Funding: No funding source
Conflicts of Interest: All authors have no conflict of interest
Acknowledgement/Disclaimers: None
References
- 1. Khorana, A., Francis, C. Risk prediction of cancer-associated thrombosis: Appraising the first decade and developing the future. (2018) Thrombosis Res 164(Suppl 1): S70-S76.
- 2. Falanga, A., Vignoli, A. Venous thromboembolism in oncology. (2004) Eksp Onkol 26(1):11-14.
PubMed│CrossRef│Others
- 3. Rickles, F., Levine, M. Epidemiology of Thrombosis in Cancer. (2001) Acta Haematologica 106(1-2): 6-12.
- 4. Lee, A., Levine, M.N. Venous Thromboembolism and Cancer: Risks and Outcomes. (2003) Circulation 107(90231): 17I--121.
- 5. Zanetto, A., Campello, E., Spiezia, L., et al. Cancer-Associated Thrombosis in Cirrhotic Patients with Hepatocellular Carcinoma. (2018) Cancers 10(11): 450.
- 6. Mulder, F., Candeloro, M., Kamphuisen, P., et al. The Khorana score for prediction of venous thromboembolism in cancer patients: a systematic review and meta-analysis. (2019) Haematologica 104(6):1277-1287.
- 7. van Es, N., Di Nisio, M., Cesarman, G., et al. Comparison of risk prediction scores for venous thromboembolism in cancer patients: a prospective cohort study. (2017) Haematologica 102(9):1494-1501.
- 8. Key, N., Khorana, A., Kuderer, N., et al. Venous Thromboembolism Prophylaxis and Treatment in Patients With Cancer: ASCO Clinical Practice Guideline Update. (2020) J Clin Oncol 38(5): 496-520.
PubMed│CrossRef│Others
- 9. Riess, H., Habbel, P., Jühling, A., et al. Primary prevention and treatment of venous thromboembolic events in patients with gastrointestinal cancers - Review. (2016) World J Gastrointest Oncol 8(3): 258-270.
- 10. Yust-Katz, S., Mandel, J., Wu, J., et al. Venous thromboembolism (VTE) and glioblastoma. (2015) J Neuro-Oncol 124(1): 87-94.
- 11. Pabinger, I., van Es, N., Heinze, G., et al. A clinical prediction model for cancer-associated venous thromboembolism: a development and validation study in two independent prospective cohorts. (2018) The Lancet Haematology 5(7): e289-e298.
- 12. Ay, C., Dunkler, D., Marosi, C., et al. Prediction of venous thromboembolism in cancer patients. (2010) Blood 116(24): 5377-5382.
- 13. Khorana, A., Francis, C., Culakova, E., et al. Risk factors for chemotherapy-associated venous thromboembolism in a prospective observational study. (2005) Cancer 104(12): 2822-2829.
- 14. Khorana, A., Vadhan-Raj, S., Kuderer, N., et al. Rivaroxaban for Preventing Venous Thromboembolism in High-Risk Ambulatory Patients with Cancer: Rationale and Design of the CASSINI Trial. (2017) Thrombosis and Haemostasis 117(11): 2135-2145.
- 15. Blom, J. Malignancies, Prothrombotic Mutations, and the Risk of Venous Thrombosis. (2005) JAMA 293(6):715.
- 16. Heit, J., O’Fallon, W., Petterson, T., et al. Relative Impact of Risk Factors for Deep Vein Thrombosis and Pulmonary Embolism. (2002) Arch Internal Med 162(11): 1245.
- 17. Heit, J., Silverstein, M., Mohr, D., et al. Risk Factors for Deep Vein Thrombosis and Pulmonary Embolism. (2000) Arch Internal Med 160(6): 809.
- 18. Couban, S., Goodyear, M., Burnell, M., et al. Randomized Placebo-Controlled Study of Low-Dose Warfarin for the Prevention of Central Venous Catheter–Associated Thrombosis in Patients With Cancer. (2005) J Clin Oncol 23(18): 4063-4069.
- 19. Akl, E., Kahale, L., Hakoum, M., et al. Parenteral anticoagulation in ambulatory patients with cancer. (2017) Cochrane Database of Syst Rev 9(9): CD006652
- 20. Agnelli, G., Gussoni, G., Bianchini, C., et al. Nadroparin for the prevention of thromboembolic events in ambulatory patients with metastatic or locally advanced solid cancer receiving chemotherapy: a randomised, placebo-controlled, double-blind study. (2009) The Lancet Oncol 10(10): 943-949.
- 21. Agnelli, G., George, D., Kakkar, A., et al. Semuloparin for Thromboprophylaxis in Patients Receiving Chemotherapy for Cancer. (2012) N Engl J Med 366(7): 601-609.
- 22. Khorana, A., Francis, C., Kuderer, N., et al. Dalteparinthromboprophylaxis in cancer patients at high risk for venous thromboembolism: A randomized trial. (2017) Thromb Res 151: 89-95.
- 23. Fagarasanu, A., Alotaibi, G., Hrimiuc, R., et al. Role of Extended Thromboprophylaxis After Abdominal and Pelvic Surgery in Cancer Patients: A Systematic Review and Meta-Analysis. (2016) Ann Surg Oncol 23(5): 1422-1430.
- 24. Verso, M., Agnelli, G., Barni, S., et al. A modified Khorana risk assessment score for venous thromboembolism in cancer patients receiving chemotherapy: the Protecht score. (2012) Intern Emerg Med 7(3): 291-292.
- 25. Khorana, A., Francis, C., Kuderer, N., et al. Dalteparin Thromboprophylaxis in Cancer Patients at High Risk for Venous Thromboembolism: A Randomized Trial. (2015) Blood 126(23): 427-427.
PubMed│CrossRef│Others
- 26. Khorana, A., Soff, G., Kakkar, A., et al. Rivaroxaban for Thromboprophylaxis in High-Risk Ambulatory Patients with Cancer. (2019) N Engl J Med 380(8): 720-728.
- 27. Carrier, M., Abou-Nassar, K., Mallick, R., et al. Apixaban to Prevent Venous Thromboembolism in Patients with Cancer. (2019) N Engl J Med 380(8): 711-719.
- 28. Agnelli, G. Direct Oral Anticoagulants for Thromboprophylaxis in Ambulatory Patients with Cancer. (2019) N Engl J Med 380(8): 781-783.
- 29. Vadhan-Raj, S., McNamara, M.G., Venerito, M., et al. Rivaroxabanthromboprohylaxis in ambulatory patients with pancreatic cancer: Results from a prespecified subgroup analysis of the CASSINI study. (2019) JCO 37(15 Suppl): 4016.
PubMed│CrossRef│Others
- 30. Key, N.S., Khorana, A.A., Kuderer, N.M., et al. Venous Thromboembolism Prophylaxis and Treatment in Patients With Cancer: ASCO Clinical Practice Guideline Update. (2020) JCO 38(5): 496-520.
- 31. Kuderer, N.M., Poniewierski, M.S., Culakova, E., et al. Predictors of Venous Thromboembolism and Early Mortality in Lung Cancer: Results from a Global Prospective Study (CANTARISK). (2018) Oncologist 23(2): 247-255.












