Assessment of Anti-Ageing Potential of Consciousness Energy Healing Treated DMEM and HFF-1 Cells using Cellular Proliferation and Collagen Metabolism Assays
Mahendra Kumar Trivedi, Alice Branton, Dahryn Trivedi, Gopal Nayak, Sambhu Charan Mondal
Affiliation
1Trivedi Global, Inc., Henderson, Nevada, USA
2Trivedi Science Research Laboratory Pvt. Ltd., Bhopal, Madhya Pradesh, India
Corresponding Author
Snehasis Jana, Trivedi Science Research Laboratory Pvt. Ltd., Bhopal, Madhya Pradesh, India. E-mail ID: publication@trivedieffect.com
Citation
Trivedi, MK., et al. Assessment of Anti-Ageing Potential of Consciousness Energy Healing Treated DMEM and HFF-1 Cells using Cellular Proliferation and Collagen Metabolism Assays. (2019) Lett Health Biol Sci 4(1): 1-6.
Copy rights
© 2019 Trivedi, MK. This is an Open access article distributed under the terms of Creative Commons Attribution 4.0 International License.
Keywords
Consciousness energy healing; The trivedi effect®; Anti-aging; HFF-1 cell line; Fibroblast cells; Collagen
Abstract
Skin health and aging are the complex biological process influenced by several intrinsic (or endogenous) and extrinsic (or exogenous) factors. Various skin-based therapies are currently available to rejuvenate the skin, but they might be related with some side-effects such as scarring. The objective of the present study was to evaluate the effect of Consciousness Energy Healing Treatment (The Trivedi Effect®) on the human foreskin fibroblast (HFF-1) cell line and Dulbecco’s Modified Eagle Medium (DMEM) for skin health parameters like cell proliferation and synthesis of collagen. The rate of cellular proliferation in HFF-1 cells was identified, and the results found that the Biofield Energy Treated DMEM significantly (p ≤ 0.001) increased by 152.38% compared to the negative control group. Additionally, the cell proliferation was also significantly increased by 71.43% in the Biofield Energy Treated cells compared to the negative control group. Similarly, the collagen level was significantly (p ≤ 0.001) increased by 60.42% in the Biofield Energy Treated DMEM compared with the negative control group. Hence, the results exhibited a significant improvement of collagen synthesis and cellular proliferation in the Biofield Energy Treated DMEM for improving skin health. It can be concluded that The Trivedi Effect® - Consciousness Energy Healing Treatment might be a complementary and alternative approach with respect to the skin health, anti-aging in DMEM compared with the HFF-1 cell line. Therefore, the Biofield Energy Treated DMEM could be useful for the development of effective cosmetic products for the prevention and treatment of several skin problems such as erythema, contact dermatitis, skin aging, wrinkles, etc.
Introduction
Skin is the largest organ in the body and plays as an important functional and psychological function. Skin aging has long been considering as an aesthetic problem than a real functional health problem. Nowadays, more peoples are reaching to old-age[1-3]. Loss of skin color, texture, and function might be responsible for aging. Aging and loss of skin function result in infection and chronic wounds[4]. Skin aging is the result of both intrinsic and extrinsic factors. Intrinsic factors are basically genetically determined, lead to chronologic aging such as fine wrinkles, skin thinning, laxity, and loss of elasticity and oxidative stress[5-7]. Extrinsic factors such as light exposure, chemicals, pollution, ionizing radiation, toxins, etc. are comes from the outside, to enhance the intrinsic skin aging and thus lead to profound wrinkles, pigmentary defect, and skin cancers[8]. Both of these factors are responsible for structural and physiological alterations in skin layers. Most of the skin manifestations are influenced by decrease in collagen level, reduced skin elasticity, atrophy, and gradual bone resorption that lead to wrinkled and dry skin. A report suggests that more than two crores skin rejuvenation procedures cases were performed in the United States in 2013[9]. To eliminate these skin difficulties, many different treatment approaches have been practiced world-wide. In addition, various photo aging products, antioxidants, pharmacological agents that have anti-aging properties (i.e. vitamin B3, C, and E), many invasive procedures like gene therapy, chemical peels, and several devices such as laser energy, injectable, and many more were used for skin health and rejuvenation[10,11]. However, various study data demonstrated that these procedures does not ensure a natural skin look and very optional, as it was found that most of the techniques are applied without any clear understanding of the skin anatomy and physiology of skin aging. Complementary and Alternative Medicine (CAM) systems are widely used against various therapeutic aspects along with skin treatment or wound repair therapies[12].
Biofield Energy Healing is one of the CAM reported with significant outcomes in clinical and pre-clinical studies. In addition to, National Center for Complementary and Alternative Medicine (NCCAM), defined Biofield Therapies under the subcategory of Energy Therapies. Biofield Energy Therapies have been reported with significant outcomes in case of arthritis patient[13], cancer patient[14], pain and anxiety cases[15], wound healing[16], and many other clinical studies with recommendations[17]. Human Biofield is a cumulative outcome of measurable magnetic and electric fields, exerted by the human body[18]. Mr. Mahendra Kumar Trivedi’s unique Biofield Energy (The Trivedi Effect®) has been scientifically studied and reported with significant outcomes in living organisms and nonliving materials in a different manner. The results of The Trivedi Effect® have been reported in the field of microbiology[19-22], agriculture[23,24], livestock[25], pharmaceutical sciences[26-29], and materials sciences[30-33]. With the increased number of growing acceptance of Biofield Energy Healing Treatment as a CAM, present study was designed to evaluate the effect of Biofield Energy Treatment (The Trivedi Effect®) on HFF-1 cell line and DMEM for skin health and aging potential with respect to the cellular proliferation assay and collagen level.
Materials and Methods
Chemicals and reagents
Dulbecco’s Modified Eagle Medium (DMEM) and fetal bovine serum (FBS) were purchased from Gibco, Genex Life Sciences Pvt. Ltd., India. Ethylenediaminetetraacetic acid (EDTA), trypsin, L-ascorbic acid and NaHCO3 were purchased from Sigma, USA. Antibiotics solution (penicillin and streptomycin) was procured from Hi-Media Pvt. Ltd., USA. Dimethyl sulphoxide (DMSO) was obtained from Thermo Fisher Scientific, USA. All the other chemicals used in this experiment were analytical grade procured from India.
Cell culture
HFF-1 (human foreskin fibroblast) cells were procured from American Type Culture Collection (ATCC), SCRC-1041™, and USA, originated from normal human skin fibroblast cells. HFF-1 cells were maintained in the growth medium (DMEM) supplemented with 15% FBS, with added antibiotics like penicillin (100 U/mL) and streptomycin (100 μg/mL). The growth condition of cell lines was at 37 ºC, 5% CO2, and 95% humidity. The cells were sub-cultured by trypsinisation followed by splitting the cell suspension into fresh flasks and supplementing with fresh cell growth medium. L-ascorbic acid and L-carnosine (positive controls) were diluted in DMEM to achieve the working concentration corresponding to the cell plate.
Consciousness energy healing treatment strategies
An aliquot of HFF-1 cells in a T-25 cell culture flask and an aliquot of culture DMEM were received the Consciousness Energy Healing (The Trivedi Effect®) Treatment by Mahendra Kumar Trivedi under standard laboratory conditions for the period of ~3 minutes from a distance of ~25 cm. The energy transmission was done without touching the cells and DMEM. Following Biofield Energy Treatment, the medium and the cells were used for the estimation of in vitro cell proliferation and collagen synthesis. The Biofield Energy Treated and untreated T-25 flask were incubated till one week in a CO2 incubator at 37 °C, 5% CO2, and 95% humidity. Besides, the Biofield Energy Treated and untreated DMEM were stored at 4ºC till cell culture.
Experimental design
Group 1 was served as the baseline control (untreated cells in untreated medium). Group 2 was defined as the negative control (untreated cells in untreated medium with H2O2). Group 3 served as positive controls (L-ascorbic acid and carnosine), i.e. cells in DMEM with ascorbic acid (10 µM) and carnosine (10 µM). Group 4 was referred as the untreated HFF-1 cells in the Biofield Energy Treated DMEM. Group 5 was served as the Biofield Energy Treated HFF-1 cells in untreated DMEM. Media was changed with 1000 µL of Biofield Energy Treated DMEM.
Cell proliferation assay
The HFF-1 cells were trypsinized, counted, and plated in 96-well plates at density of 5 X 103 cells/well/180 µL of growth medium followed by overnight incubation for cell recovery and exponential growth. Further, the cells were subjected to serum starvation so as to synchronize cell growth. These cells were treated as per experimental procedure with positive controls (ascorbic acid and carnosine) and the test item (Biofield Energy Treated cells and DMEM) followed by the incubation for 72 hours in a CO2 incubation at 37 °C, 5% CO2, and 95% humidity. About 20 µL of 5 mg/mL of MTT 3-(4,5-dimethythiazol-2-yl)-2,5-diphenyl tetrazolium bromide solution was added followed by additional incubation for 3 hours at 37 °C. The supernatant was aspirated and 150 µL of DMSO was added to each well to dissolve the formazan crystals followed by measurement of absorbance at 540 nm using Synergy HT microplate reader[3,4].
Estimation of collagen synthesis
The HFF-1 cells were trypsinized, counted and plated in wells of 48-well plates at a density corresponding to 10 X 103 cells/well/0.5 mL of cell growth medium followed by overnight incubation. Further, the cells were subjected to serum starvation in order to synchronize the cell growth. These cells were treated with the positive controls and the test samples i.e., the Biofield Energy Treated cells and DMEM followed by incubation for 72 hours at 37 °C, 5% CO2, and 95% humidity. After incubation, the plates were removed and the amount of collagen accumulated in HFF-1 cells corresponding to each treatment groups were measured by direct Sirius red dye binding assay[35,36].
Statistical analysis
Data were expressed as mean of three replicates ± standard deviation (SD) and were subjected to one-way analysis of variance (ANOVA) with Dunnett’s post-hoc test using SigmaPlot statistical software version 11.0. Statistical significance was considered at p<0.05.
Results and Discussion
Cellular proliferation assay
Effect of the Biofield Energy Treatment on DMEM and HFF-1 cells for cellular proliferation is shown in Table 1. After 72 hours of incubation the positive controls like L-ascorbic acid and carnosine were significantly increased the cell growth by 16.92% and 7.69%, respectively in HFF-1 cells compared with the negative control group (Table 1). Similarly, the Biofield Energy Treated DMEM and HFF-1 cells showed significant (p≤ 0.001) changed in absorbance i.e. 0.53 ± 0.03 and 0.36 ± 0.02, respectively, indicating a significant increase in cellular proliferation. The cell proliferation assay was used to assess the effects of pharmacological agents or growth factors, cytotoxic potential or investigate the circumstances of cell activation. In this assay, the numbers of cells change in the proportion of cells that are in dividing state were considered. There are different types of cell proliferation assays, depending on the actual needs of measurement such as DNA synthesis, metabolic activity, antigens associated with cell proliferation and ATP concentration. Cell health is depends on the rate of cell proliferation or viability. Proliferation or viability analysis is very crucial for the studies of cell growth and differentiation. Proliferation is also considered as a convenient measure of population dynamics in studies of cytokines or growth factors[37,38].
Table 1: Effect of the Biofield Energy Treated DMEM and HFF-1 cells on cellular proliferation
|
Group |
Description |
Absorbance (540 nm) |
|
Baseline (0 hour) |
Cells + DMEM |
0.17 ± 0.01 |
|
Baseline (72 hours) |
Cells + DMEM |
0.23 ± 0.01 |
|
Negative control |
Cells + DMEM + H2O2 (200 µM) |
0.21 ± 0.001 |
|
Positive control |
Cells + DMEM + L-Ascorbic acid (10 µM) + H2O2 (200 µM) |
0.22 ± 0.004 |
|
Cells + DMEM + L-Carnosine + H2O2 (200 µM) |
0.21 ± 0.00 |
|
|
Biofield Energy Treated DMEM |
Cells + Treated DMEM + H2O2 (200 µM) |
0.53 ± 0.03*** |
|
Biofield Energy Treated Cells |
Treated cells + DMEM + H2O2 (200 µM) |
0.36 ± 0.02*** |
Each value represents as mean ± SD of triplicate wells. Statistical comparison was performed using one-way analysis of variance (ANOVA) with Dunnett’s post-test. ***p≤ 0.001 with respect to the negative control group.
The results of cell proliferation in all the experimental groups are represented in Figure 1. The baseline control group at 72 hours (equivalent to non-senescent cells) on the basis of respective absorbance was tabulated as 100%, while the negative control group (equivalent to aged/senescent cells) showed significantly decreased by 35% in proliferation rate. The positive controls like ascorbic acid and carnosine showed an increased the rate of cell proliferation by 11% and 5%, respectively compared to the negative control group. The Biofield Energy Treated DMEM led to a significantly (p≤ 0.001) increased in cell proliferation of HFF-1 cells compared to cells treated with H2O2 alone as well as non-senescent cells as indicated by 472% higher proliferation over the untreated i.e. non-senescent cells. Further, the Biofield Energy Treated cells demonstrated a significantly (p≤0.001) increased in cell proliferation compared to cells treated with H2O2 alone as well as non-senescent cells as indicated by 208% higher proliferation of Biofield Energy (known as The Trivedi Effect®) Treated cells over non-senescent cells. Hence, it can be concluded that the Biofield Energy Treatment (The Trivedi Effect®) on DMEM and HFF-1 cells would be observed with significant cellular proliferation. These findings suggested that the cellular proliferation rate after Biofield Energy Treatment was significantly increased. The significant enhanced proliferation rate and migration were reported in the Biofield Energy Treated DMEM as well as HFF-1 cells. Cell proliferation and migration are the important parameters, which can be utilized in skin health in terms of wound healing, skin regeneration potential, antiaging, etc.

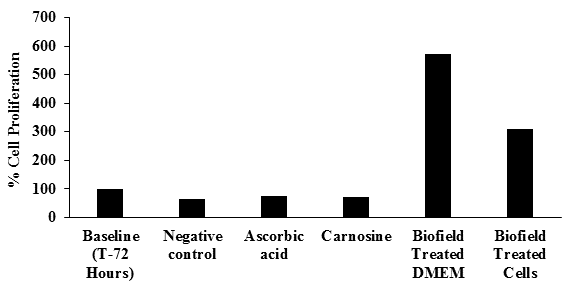
Figure 1: Effect of the test item on cell proliferation measured at 540 nm in HFF-1 cells.
Estimation of collagen level
Collagen level in the Biofield Energy Treated DMEM and HFF-1 cells was estimated using ascorbic acid and carnosine as positive controls. The results of collagen level and the percentage increase with respect to baseline control group are summarized in Table 2. Collagen is very important tissue to maintain the integrity and elasticity of the body structures. The main role of collagen is to sustain the tendons, skin, and cartilage[39]. Collagen type 1 is present in the skin structure and one of the major constituent of dermis[40]. To maintain the skin health, various amino acids have been recommended that may prevent the skin aging by increasing the dermal collagen synthesis. Many products based on supplementation of proline and its precursors such as glutamate reported with significantly increase in collagen synthesis in human dermal fibroblast cells[41]. However, as per standard data, overall dermal collagen per unit area of the skin surface is declining approximately by 1% per year[42]. The baseline control group cells in the presence of DMEM were reported with collagen 16.7 ± 0.93 µg/mL. However, after challenged with H2O2 in the negative control group the level of collagen was significantly (p≤ 0.05) decreased by 13.77% compared to the baseline control group. The positive controls ascorbic acid and carnosine showed an increased collagen concentration i.e. 15.5 ± 0.43 and 16.0 ± 1.71 µg/mL, respectively. The percentage increase in the collagen level in the ascorbic acid and carnosine groups was 7.64% and 11.11% respectively, compared with the negative control group. Similarly, the collagen level in the Biofield Energy Treated DMEM and HFF-1 cells was found as 23.10 ± 0.38 and 14.0 ± 1.45, respectively compared to the negative control group and the change was significant in the Biofield Energy Treated DMEM compared with the Biofield Energy Treated HFF-1 cells. However, the Biofield Energy Treated DMEM showed a significant (p≤ 0.001) increased the collagen level by 60.42%, while the Biofield Energy Treated cells showed 2.7% decreased in collagen content compared with the negative control group.
Table 2: Collagen level analysis in the Biofield Energy Treated HFF-1 cells and DMEM for potent anti-aging or anti-fibrotic potential
|
Group |
Description |
Collagen (µg/mL) at 540 nm |
% Collagen change |
|
Baseline (72 hours) |
Cells + DMEM |
16.7 ± 0.93 |
- |
|
Negative control |
Cells + DMEM + H2O2 (200 µM) |
14.4 ± 0.52 |
-13.77* |
|
L-Ascorbic acid |
Cells + DMEM + L-Ascorbic acid + H2O2 (200 µM) |
15.5 ± 0.43 |
7.64 |
|
L-Carnosine |
Cells + DMEM + L-Carnosine + H2O2 (200 µM) |
16.0 ± 1.71 |
11.11 |
|
Biofield Energy Treated DMEM |
Cells + Treated DMEM + H2O2 (200 µM) |
23.10 ± 0.38 |
60.42*** |
|
Biofield Energy Treated cells |
Treated Cells + DMEM + H2O2 (200 µM) |
14.0 ± 1.45 |
-2.7 |
Each value represents mean ± SD of triplicates wells. Statistical comparison with the untreated group was conducted using one-way analysis of variance (ANOVA) with Dunnett’s post-test. *p≤ 0.05 with respect to the baseline control group (using Student’s t-test); ***p≤ 0.001 with respect to the negative control group.
Experimental data suggests that vitamin C (ascorbic acid) stimulated the formation of skin collagen possibly by increased epidermal moisture content, and improving the skin hydration tendency[43]. Besides, carnosine stimulates the process of wound healing through the stimulation of histamine and collagen biosynthesis and increased granulation[44]. In this experiment, the Biofield Energy Treated DMEM significantly improved the collagen level, which could be useful in improving skin health, anti-ageing, improves skin hydration capacity, etc. Overall, it can be concluded that The Trivedi Effect® can improve the cellular proliferation rate and collagen content, which would be directly related to maintaining the skin health. The Consciousness Energy Healing Treatment, an energetic matrix surrounds the human body, also known as complex, dynamic, weak energy field. It might be expected that the Biofield Energy Treatment might control the oxidative damage to the skin membrane, maintain local pH changes results in improved the cell proliferation and collagen content. It can be assumed that The Trivedi Effect® can be used as a potent anti-aging or anti-fibrotic agent to promote the skin health.
Conclusion
The percent cellular proliferation was significantly (p≤ 0.001) increased by 152.38% and 71.43% in the Biofield Energy Treated DMEM group and Biofield Energy Treated HFF-1 cell group, respectively compared with the baseline control group. Additionally, the level of collagen was significantly (p≤ 0.001) increased by 60.42% in the Biofield Energy Treated DMEM group compared with the negative control group. However, the Biofield Energy Treated HFF-1 cells did not show any significant difference in collagen level. Hence, it can be concluded that the Consciousness Energy Healing Treatment (The Trivedi Effect®) has the potential to have significant action as wound healing, antiaging, and skin regeneration and as an alternative method to improve the skin health and it would be useful for the management of wounds and various skin-related disorders viz. skin abscess, pimples, cellulitis, impetigo, scabies, syringoma, photosensitivity, urticaria, hives, warts, abscess, callus, acne, chickenpox, eczema, rosacea, seborrheic dermatitis, athlete’s foot, psoriasis, erythema, contact dermatitis, cutis rhomboidalis nuchae, etc.
Acknowledgements: Authors gratefully acknowledged to Trivedi Science, Trivedi testimonials and Trivedi master wellness for their support. In addition, authors are thankful for the support of Dabur Research Foundation for conducting this study.
References
- 1. United Nations, Department of Economic and Social Affairs, Population Division. (2013) World Population Ageing 2013. ST/ESA/SER.A/348.
Pubmed | Crossref | Others
- 2. United Nations, Department of Economic and Social Affairs, Population Division World Population Prospects: The 2015 Revision, Key Findings and Advance Tables. (2015) Working Paper No. ESA/P/WP.241.
Pubmed | Crossref | Others
- 3. Jamison, D.T., Summers, L.H., Alleyne, G., et al. Global health 2035: A world converging within a generation. (2013) Lancet 382: 1898-1955.
- 4. Ganceviciene, R., Liakou, A.I., Theodoridis, A., et al. Skin anti-aging strategies. (2012) Dermatoendocrinol 4: 308-319.
- 5. Farage, M.A., Miller, K.W., Elsner, P., et al. Intrinsic and extrinsic factors in skin ageing: A review. (2008) Int J Cosmet Sci 30(2): 87-95.
- 6. Kammeyer, A., Luiten, R.M. Oxidation events and skin aging. (2015) Ageing and skin research reviews 21: 16-29.
- 7. Rinnerthaler, M., Bischof, J., Streubel, M.K., et al. Oxidative stress in aging human skin. (2015) Biomolecules 5(2): 545-589.
- 8. Krutmann, J., Liu, W., Li, L., et al. Pollution and skin: From epidemiological and mechanistic studies to clinical implication. (2014) J Dermatol Sci 76(3): 163-168.
- 9. Loesch, M.M., Somani, A.K., Kingsley, M.M., et al. Skin resurfacing procedures: New and emerging options. (2014) Clin Cosmet Investig Dermatol 7: 231-241.
- 10. Baumann, L. Skin ageing and its treatment. (2007) J Pathol 211(2): 241-251.
- 11. Uitto, J. Understanding premature skin aging. (1997) N Engl J Med 337(20): 1463-1465.
- 12. Dorai, A.A. Wound care with traditional, complementary and alternative medicine. (2012) Indian J Plast Surg 45: 418-424.
- 13. Peck, S.D. The efficacy of therapeutic touch for improving functional ability in elders with degenerative arthritis. (1998) Nurs Sci Q 11: 123-132.
- 14. Giasson, M., Bouchard, L. Effect of therapeutic touch on the well-being of persons with terminal cancer. (1998) J Holist Nurs 16(3): 383-398.
- 15. Turner, J.G., Clark, A.J., Gauthier, D.K., et al. The effect of therapeutic touch on pain and anxiety in burn patients. (1998) J Adv Nurs 28(1): 10-20.
- 16. Schlitz, M., Hopf, H.W., Eskenazi, L., et al. Distant healing of surgical wounds: An exploratory study. (2012) Explore (NY) 8: 223-230
- 17. Jain, S., Hammerschlag, R., Mills, P., et al. Clinical studies of biofield therapies: Summary, methodological challenges, and recommendations. (2015) Glob Adv Health Med 4: 58-66.
- 18. Movaffaghi, Z., Farsi, M. Biofield therapies: Biophysical basis and biological regulations. (2009) Complement Ther Clin Pract 15(1): 35-37, 31.
- 19. Trivedi, M.K., Patil, S., Shettigar, H., et al. Antimicrobial sensitivity pattern of Pseudomonas fluorescens after biofield treatment. (2015) J Infect Dis Ther 3: 222.
Pubmed | Crossref | Others
- 20. Trivedi, M.K., Patil, S., Shettigar, H., et al. Phenotypic and biotypic characterization of Klebsiella oxytoca: An impact of biofield treatment. (2015) J Microb Biochem Technol 7: 203-206.
Pubmed | Crossref | Others
- 21. Trivedi, M.K., Patil, S., Shettigar, H., et al. Effect of biofield treatment on phenotypic and genotypic characteristic of Provindencia rettgeri. (2015) Mol Biol 4: 129.
Pubmed | Crossref | Others
- 22. Patil, S.A., Nayak, G.B., Barve, S.S., et al. Impact of biofield treatment on growth and anatomical characteristics of Pogostemon cablin (Benth.). (2012) Biotechnology 11(3): 154-162.
Pubmed | Crossref | Others
- 23. Nayak, G., Altekar, N. Effect of biofield treatment on plant growth and adaptation. (2015) J Environ Health Sci 1: 1-9.
Pubmed | Crossref | Others
- 24. Shinde, V., Sances, F., Patil, S., et al. Impact of biofield treatment on growth and yield of lettuce and tomato. (2012) Aust J Basic Appl Sci 6(10): 100-105.
Pubmed | Crossref | Others
- 25. Trivedi, M.K., Branton, A., Trivedi, D., et al. Effect of biofield treated energized water on the growth and health status in chicken (Gallus gallus domesticus). (2015) Poult Fish Wildl Sci 3: 140.
Pubmed | Crossref | Others
- 26. Trivedi, M.K., Branton, A., Trivedi, D., et al. Gas Chromatography-mass spectrometric analysis of isotopic abundance of 13C, 2H, and 18O in biofield energy treated p-tertiary butylphenol (PTBP). (2016) American J Chemical Engineering 4(4): 78-86.
Pubmed | Crossref | Others
- 27. Trivedi, M.K., Branton, A., Trivedi, D., et al. Gas chromatography-mass spectrometry based isotopic abundance ratio analysis of biofield energy treated methyl-2-napthylether (Nerolin). (2016) American Journal of Physical Chemistry 5: 80-86.
- 28. Trivedi, M.K., Branton, A., Trivedi, D., et al. Isotopic abundance ratio analysis of 1,2,3-trimethoxybenzene (TMB) after biofield energy treatment (The Trivedi Effect®) using gas chromatography-mass spectrometry. (2016) American Journal of Applied Chemistry 4(4): 132-140.
Pubmed | Crossref | Others
- 29. Trivedi, M.K., Branton, A., Trivedi, D., et al. Determination of isotopic abundance ratio of biofield energy treated 1,4-dichlorobenzene using gas chromatography-mass spectrometry (GC-MS). (2016) Modern Chemistry 4(3): 30-37.
Pubmed | Crossref | Others
- 30. Trivedi, M.K., Tallapragada, R.M., Branton, A., et al. The potential impact of biofield energy treatment on the atomic and physical properties of antimony tin oxide nanopowder. (2015) American Journal of Optics and Photonics 3: 123-128.
Pubmed | Crossref | Others
- 31. Trivedi, M.K., Tallapragada, R.M., Branton, A., et al. Characterization of physical and structural properties of aluminium carbide powder: Impact of biofield treatment. (2015) J Aeronaut Aerospace Eng 4: 142.
Pubmed | Crossref | Others
- 32. Trivedi, M.K., Nayak, G., Patil, S., et al Impact of biofield treatment on atomic and structural characteristics of barium titanate powder. (2015) Ind Eng Manage 4: 166.
Pubmed | Crossref | Others
- 33. Trivedi, M.K., Tallapragada, R.M., Branton, A., et al. Evaluation of physical and structural properties of biofield energy treated barium calcium tungsten oxide. (2015) Advances in Materials 4: 95-100.
Pubmed | Crossref | Others
- 34. Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. (1983) J Immunol Meth 65(1-2): 55-63.
- 35. Junqueira, L.C., Bignolas, G., Brentani, R.R. Picrosirius staining plus polarization microscopy, a specific method for collagen detection in tissue sections. (1979) Histochem J 11(4): 447-455.
- 36. Junquiera, L.C., Junqueira, L.C., Brentani, R.R. A simple and sensitive method for the quantitative estimation of collagen. (1979) Anal Biochem 94(1): 96-99.
- 37. Yadav, K., Singhal, N., Rishi, V., et al. Cell Proliferation Assays. (2014) In: eLS. John Wiley & Sons Ltd.
Pubmed | Crossref | Others
- 38. Roederer, M. Interpretation of cellular proliferation data: Avoid the panglossian. (2011) Cytometry A 79(2): 95-101.
- 39. Kligman, L.H. Photoaging. Manifestations, prevention, and treatment. (1989) Clin Geriatr Med 5(1): 235-251.
- 40. Takema, Y., Hattori, M., Aizawa, K. The relationship between quantitative changes in collagen and formation of wrinkles on hairless mouse skin after chronic UV irradiation. (1996) J Dermatol Sci 12(1): 56-63.
- 41. Karna, E., Miltyk, W., Wolczynski, S., et al. The potential mechanism for glutamine-induced collagen biosynthesis in cultured human skin fibroblasts. (2001) Comp Biochem Physiol B Biochem Mol Biol 130(1): 23-32.
- 42. Shuster, S., Black, M.M., McVitie, E. The influence of age and sex on skin thickness, skin collagen and density. (1975) Br J Dermatol 93(6): 639-643.
- 43. Fusco, D., Colloca, G., Lo Monaco, M.R., et al. Effects of antioxidant supplementation on the aging process. (2007) Clin Interv Aging 2(3): 377-387.
- 44. Nagai, K., Suda, T., Kawasaki, K., et al. Action of carnosine and beta-alanine on wound healing. Surgery 100(5): 815-821.












