Giant Primary Ovarian Leiomyoma- A Diagnostic Dilemma
Vidhya Manohar1, Vishwas. D. Pai2*, Sunitha. S. Kerudi3, Suvarna Ravindranath1, Satish Tumbal4
Affiliation
- 1Department of Pathology, SRL Diagnostics, Banagalore, Karnataka, India
- 2Department of Surgical Oncology, Kerudi Cancer Hospital, Bagalkot, Karnataka, India
- 3Department of Gynecology, Kerudi Hospital, Bagalkot, Karnataka, India
- 4Department of Radiodiagnosis, Kerudi Cancer Hospital, Bagalkot, Karnataka, India
Corresponding Author
Vishwas. D. Pai, Department of Surgical Oncology, Kerudi Cancer Hospital, Bagalkot, Karnataka, India: 587101, Tel: +91-9449333502; E-mail: vishpai88@gmail.com
Citation
Vishwas, D.P., et al. Giant Primary Ovarian Leiomyoma – A Diagnostic Dilemma. (2016) J Anesth Surg 3(2): 148-150.
Copy rights
© 2016 Vishwas, D.P. This is an Open access article distributed under the terms of Creative Commons Attribution 4.0 International License
Keywords
Leiomyoma; Ovarian benign tumour
Introduction
Leiomyomas are rare solid tumours of the ovary accounting for 0.5 - 1% of the benign ovarian tumours[1]. Usually these tumours occur in the reproductive age group and affect both ovaries[2,3]. Majority of these tumours are detected either during routine physical examination or incidentally during surgery or autopsy[4]. Owing to the rarity and similar age of presentation, they are frequently misdiagnosed as germ cell tumours particularly when large in size. We are presenting a case of giant ovarian leiomyoma in a young lady which was suspected as germ cell tumour on imaging.
Case Report
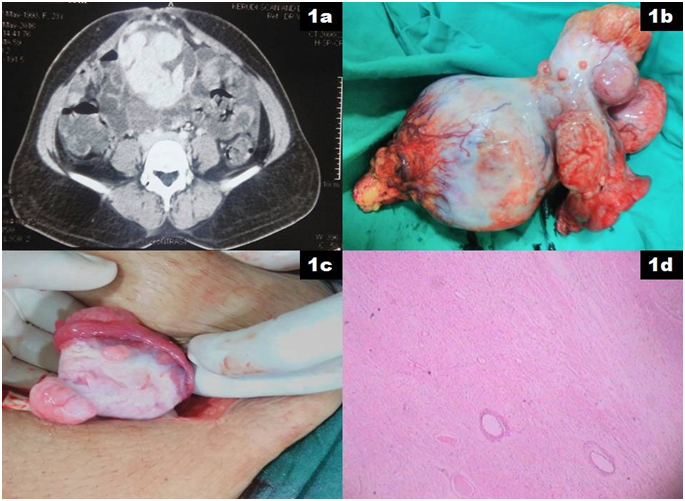
A 23 years old lady, mother of a single child presented with history of irregular menstruation of 6 months duration. Abdominal examination revealed 10 X 8 cm hard, well defined mass arising from pelvis which was mobile in both horizontal and vertical direction. She was evaluated with ultrasound examination of the abdomen which revealed 12 X 10 X 8 cm solid, heterogeneous mass arising from right adnexa and 3 X 2 cm homogenous solid mass arising from left adnexa. Tumour markers including CA 125, AFP (Alfa feto protein), B-HCG (Beta human chorionic gonadotrophin) and LDH (Lactate Dehydrogenase) were within normal limits. Contrast enhanced computed tomography (CECT) of the abdomen revealed 13 X 13 X 11 cm lobulated mixed (predominantly solid) markedly enhancing soft tissue mass is noted in the right adnexal region with enhancing intralesional and perilesional vessels with peripheral irregular dense calcification suggestive of yolk sac tumour (Figure 1a). A similar lesion of size 4 X 3 X 2 cm was noted in the left adnexal region. In view of imaging findings, normal levels of tumour markers, young age and incomplete family, tentative plan of treatment was right salpingo-oophorectomy and decision on chemotherapy based on histopathological report. Right salpingo-oophorectomy with inspection of the left ovarian lesion was done (Figure 1b and 1c). Surgical staging in the form of cytology of the peritoneal fluid, omental as well as peritoneal biopsy were performed. Intra and post operative course were uneventful and patient was discharged on the 1st post operative day. Histopathological examination revealed tumour composed of interlacing bundles of smooth muscles with oedematous intervening stroma suggestive of leiomyoma (Figure 1d). Patient was kept under observation for the left ovarian leiomyoma. Her menstrual cycles regularized after the surgery and at last follow up, she was in good health.
Figure 1a: CECT abdomen showing mixed, markedly enhancing soft tissue mass arising in the right adnexal region with enhancing intralesional and perilesional vessels with peripheral irregular dense calcification suggestive of yolk sac tumour.
Figure 1b: Right salpingo-oophorectomy specimen showing well defined, irregular, hard mass arising from the ovary and involving the fallopian tube.
Figure 1c: Intra operative picture of the left ovary showing small, well defined, hard mass arising from the left ovary.
Figure 1d: Microscopic examination of the resected tumour composed of interlacing bundles of smooth muscles with oedematous intervening stroma suggestive of leiomyoma.
Discussion
Primary leiomyoma of the ovary (PLO) is rare with less than 100 cases reported in English literature[5,6]. However it is difficult to estimate the exact incidence as most of these lesions remain asymptomatic and are detected incidentally during surgery[4]. PLO predominantly affects women of reproductive age although few cases have been reported in the post menopausal age group. It has been observed that PLO occurring in the reproductive age group is predominantly bilateral whereas those affecting the post menopausal age group are predominantly unilateral[7]. Present patient was a young lady and both ovaries were affected.
Exact pathogenesis of this rare entity remains to be established. Although a number of theories have been proposed, the most accepted one is that the tumour arises from the smooth muscle cells of the ovarian hilar blood vessels[8]. Other probable sites of origin include cells in the ovarian ligament, smooth muscle cells, multipotent cells in the ovarian stroma or undifferentiated germ cells in the ovarian stroma[9]. Endometriotic cysts which may trigger metaplasia of the surrounding stroma into the smooth muscle cells may also result in the development of PLO[10]. Additionally, metaplasia of smooth muscles present in mature cystic teratomas or mucinous cystic tumour may explain the pathogenesis in some cases.
In general, PLOs are small measuring only few millimetres or centimetres in diameter. Herbut et al[11] had reported a huge PLO of about 25 cm in diameter. Similarly Agarwal et al[4] had reported a PLO of size more than 20 cm. In the present patient PLO of the right ovary was 13 cm X 13 cm X 11 cm in diameter. Majority of these tumours are asymptomatic due to small size of the tumours. When symptomatic, they are related to the presence of the adnexal mass in the form of abdominal pain or swelling. Rarely the presentation is acute with torsion of the ovarian mass with resultant haemorrhage and necrosis. In general, menstrual irregularities are rare in PLO. Present patient presented with irregular menstrual cycles which triggered further investigations leading to the diagnosis of PLO.
On gross examination, these tumours are solid and firm with smooth surfaces. On cut-section, they have a white or greywhite surface associated with whorl formation. Secondary changes in the form of hyalinization, dystrophic calcification or cystic changes may be present in some cases[5,12]. On microscopic examination, these tumours demonstrate smooth muscle cells with elongated cigar shaped nuclei. Ideally, a PLO should be entirely within the ovary, with no similar lesions elsewhere. In the present patient, the tumour had replaced greater portion of the ovary although some normal ovarian remnant could still be identified.
Differential diagnoses include sex cord stromal tumours like the comas or fibromas, leiomyosarcoma and uterine leiomyoma becoming parasites to the ovary(wandering Leiomyomas)[6,13,14]. Immunohistochemical analysis (IHC) is essential in a number of cases to establish the diagnosis of PLO. Leiomyomas show diffuse positivity for desmin whereas fibromatous tumours are characteristically negative or only focally positive. In the present patient, diagnosis was quite apparent on microscopic examination and hence IHC analysis was avoided.
Management depends on the mode of presentation. When symptomatic or large in size or associated with complications like torsion, surgical resection is indicated whereas asymptomatic and small tumours can be observed owing to their benign nature. Present patient had giant PLO and was misdiagnosed preoperatively as germ cell tumour.
Conclusion
The purpose of presenting this case was to stress the rarity of the disease as well as the importance of proper pathological examination of all the adnexal masses.
References
- 1. Lim, S., Jeon, H. Bilateral primary ovarian leiomyoma in a young woman: case report and literature review. (2004) Gynecol Oncol 95(3): 733–735.
- 2. Safaei, A., Khanlari, M., Azarpira, N., et al. Large ovarian leiomyoma in a postmenopausal woman. (2011) Indian J Pathol Microbiol 54(2): 413–414.
- 3. Murakami, M., Uehara, H., Nishimura, M., et al. A huge ovarian smooth muscle tumor: a case report. (2010) J Med Invest 57(1-2): 158–162.
- 4. Agrawal, R., Kumar, M., Agrawal, L., et al. A huge primary ovarian leiomyoma with degenerative changes-an unusual. (2013) J Clin Diagn Res 7(6): 1152–1154.
- 5. Ramalakshmi Pullela, V.B., Das, S., Chunduru, B., et al. A case of ovarian leiomyoma with cystic degeneration. (2009) Indian J Pathol Microbiol 52(4): 592–594.
- 6. Lerwill, M.F., Sung, R., Oliva, E., et al. Smooth muscle tumors of the ovary: a clinicopathologic study of 54 cases emphasizing prognostic criteria, histologic variants, and differential diagnosis. (2004) Am J Surg Pathol 28(11): 1436–1451.
- 7. Lim, S., Jeon, H. Bilateral primary ovarian leiomyoma in a young woman: case report and literature review. (2004) Gynecol Oncol 95(3): 733–735.
- 8. Fukunaga, M. Smooth muscle metaplasia in ovarian endometriosis. (2000) Histopathology 36(4): 348–352.
- 9. Tomas, D., Lenicek, T., Tuckar, N., et al. Primary ovarian leiomyoma associated with endometriotic cyst presenting with symptoms of acute appendicitis: a case report. (2009) Diagn Pathol 4: 25.
- 10. Scully, R.E. Smooth-muscle differentiation in genital tract disorders. (1981) Arch Pathol Lab Med 105(10): 505–507.
- 11. Herbut, P.A. Ovarian leiomyoma: In Gynaecological and obstetrical pathology. (1953) Lea and Febiger, Philadelphia 1st edition: 501.
- 12. Wei, C., Lilic, N., Shorter, N., et al. Primary ovarian leiomyoma: a rare cause of ovarian tumor in adolescence. (2008) J Pediatr Adolesc Gynecol 21(1): 33–36.
- 13. Doss, B.J., Wanek, S.M., Jacques, S.M., et al. Ovarian leiomyomas: clinicopathologic features in fifteen cases. (1999) Int J Gynecol Pathol 18(1): 63–68.
- 14. Prayson, R.A., Hart, W.R. Primary smooth-muscle tumors of the ovary. A clinicopathologic study of four leiomyomas and two mitotically active leiomyomas. (1992) Arch Pathol Lab Med 116(10): 1068–1071.