Gut Microbiota and Small Intestine Bacterial Overgrowth
Wilson Daza
Affiliation
Pediatric Gastroenterologist & Master in Clinical Nutrition. Associate Professor, School Medicine, El Bosque University
Corresponding Author
Wilson Daza, Pediatric Gastroenterologist & Master in Clinical Nutrition. Associate Professor, School Medicine, El Bosque University; E-mail: dazawilson@unbosque.edu.co
Citation
Daza, W., et al. Gut Microbiota and Small Intestine Bacterial Overgrowth. (2017) J Gastro Disor liv func 3(1):114- 119.
Copy rights
© 2017 Daza, W. This is an Open access article distributed under the terms of Creative Commons Attribution 4.0 International License.
Introduction
The intestinal microbiota is made up of a complex group of microorganisms. Presently, it is considered to be a true organ with multiple metabolic, immunological, and barrier functions for the host, to the extent that there exists a symbiotic relationship between the microorganisms and the human being[1].
Composition of the intestinal microbiota
The number of bacteria that make up the microbiota is greater than the number of cells in a human being. In its composition four families of bacteria stand out: Firmicutes, Bacteroidetes, Actinobacteria, and Proteobacteria. Up to several years ago, it was estimated that a newborn had a sterile digestive system. However, the presence of microorganisms has been demonstrated in the meconium, the placenta, and the umbilical cord, which suggests the intrauterine beginning of the colonization[1]. Nevertheless, this colonization happens mainly at the postnatal stage and completes its maturation between 2 and 4 years of age. After birth, there exists a predominance of aerobic or facultative anaerobic microorganisms such as Staphylococcus, Enterococcus, and Lactobacillus, among others, which consume the intraluminal oxygen. This favors the habitat for strictly anaerobic bacteria such as Bifidobacterium, Bacteroides, and Eubacterium[1-3].
The bacterial density varies according to the locality and exponentially increases as the digestive tube advances[1,4]. In the duodenum and in the proximal jejune, gram-positive aerobics or facultative anaerobics (Enterococcus and Lactobacillus) predominate. The distal ileum is a transition zone where aerobic bacteria, typical of the small intestine, and anaerobic ones, typical of the colon, are found[1,4].
The composition and the profile of the microbiota are influenced by different factors, such as the manner of birth, maternal breastfeeding and its duration, the use of infantile formula, the age of introduction of complementary feeding, and antibiotic therapy, among others[1,4]. Some of these aspects determine whether the microbiota will be more or less healthy. Also, the intestinal microbiota can be modified including during the day by the effect of other determinants such as eating characteristics, stress, and geographical location. Some studies have demonstrated that a high-fiber diet increases the family Prevotella, while an intake high in carbohydrates increases the number of Firmicutes. For its part, a diet rich in animal proteins favors Bacteroides, with a decrease in Firmicutes[4].
Functions of the intestinal microbiota
The microbiota carries out multiple nutritional, metabolic, and immune functions, among others[1,4,6]. As for its immunological functions, the strengthening of the barrier effect of the intestinal mucus stands out, which promotes the production of mucus that lines the intestine|| Some pathogenic microorganisms adhere to this mucus and become trapped, which facilitates their elimination by means of bowel movements. Furthermore, the microbiota produces bactericidal enzymes that inhibit the proliferation of microorganisms. It also actively participates in the modulation of the immune response[6].
As part of the metabolic functions, the formation of nutrients on the basis of non-digestible compounds in the diet such as fiber, for example, and Short-Chain Fatty Acids (SCFAs) such as propionic, butyric, and acetic acid, energy for the excellence of the colonocytes (surface cells of the colon) should be mentioned. The principal activity of the colonocytes is to absorb water, although they also carry out protective functions and collaborate with the absorption of minerals such as calcium and iron, among others. In the same way, the microbiota assists in the digestion of certain carbohydrates such as lactose and participates in the synthesis of vitamins such as pyridoxine(B6) cobalamin(B12), thiamin(B1), folic acid, and nicotinic acid. This type of activity mainly depends on bacteria of the genus Bifidobacterium[1,6].
At the same time, there has been evidence that the intestinal microbiota has an impact on other organs at a distance, such as the brain, where it promotes the production of neuroendocrine substances that influence the hypothalamus-hypophysis axis, which in turn has multiple systemic effects[7]. That is the case with regulation of the appetite, a consequence of some substances derived from the metabolism of carbohydrates by the microbiota, which stimulates the secretion of the peptide YY, an intestinal hormone that suppresses the appetite and intake of food[7].
Small intestine Bacterial Overgrowth
Some pediatric illnesses have been associated with the modification of the intestinal microbiota, such as, for example, infantile colic, persistent diarrhea, irritable bowel syndrome, necrotizing enterocolitis, celiac disease, food allergy, and inflammatory bowel disease, and furthermore with some entities that extend to adult life, such as diabetes and obesity, among others[1,5].
Likewise, in patients with entities that are accompanied by inflammation, such as celiac disease and irritable bowel syndrome, the alteration of the intestinal motility raises the possibility of bacterial overgrowth, a consequence of the stasis in the intestinal content, which favors a greater number of bacteria in the intestine[1,7,8]. At the same time, some medical practices, such as treatment with proton pump inhibitors, which can cause hypochlorhydria or resection of the ileocecal valve, which allows the passage of bacteria of the colon to the small intestine, predispose an increase in the number of bacteria in the small intestine, with symptoms that are not always associated with bacterial overgrowth[1,7].
Small Intestine Bacterial Overgrowth (SIBO) syndrome is defined as an increase in the number of non-pathogenic bacteria, characterized by >105 Colony-Forming Units (CFU) /mL of bacteria in the proximal small intestine[8]. The worldwide prevalence of SIBO is unknown, because frequently it is under diagnosed. Studies have been carried out to evaluate the frequency of SIBO in specific illnesses, principally in adults[6,7,8].
The predisposing etiology for SIBO is varied. It has been associated with multiple entities and causes where the homeostatic mechanisms are altered (Table 1), among them, acquired or congenital anatomic alterations (intestinal resection, loss of the ileocecal valve, and/or enteroenteric or enterocolic fistulas), diminished gastric acid secretion (for example, secondary to the chronic use of acid suppressants or due to chronic atrophic gastritis), alterations in motility, and acquired small intestine illnesses and immune-deficiencies, primary or acquired[10]. In exocrine pancreatic insufficiency, SIBO associated with a deficit of antibacterial proteolytic enzymes with changes in motility has been observed. On the other hand, antibiotic therapy has been correlated with the appearance of SIBO, due to the modification of the composition of the intestinal microbiota, with an increase in pathogenic bacteria, which impacts digestion and the absorption of some nutrients[8].
Table 1: Causes that predispose Small Intestine Bacterial Overgrowth (SIBO) syndrome[20].
| Causes | Etiology |
| Anatomical | Enteroenteric fistulasDiverticulitis of the small intestineIntestinal stenosis Resection of the ileocecal valve Short bowel syndrome |
| Functional | Intestinal motility syndromes Hypochlorhydria or achlorhydriaInflammatory diseases: Crohn’s diseaseAutonomous Neuropathies Alteration of the lymphoid tissue associated with the intestineIrritable bowel syndrome |
| Miscellaneous | Use de acid suppressors and anti motilityPrevious antibiotic therapy Primary and/or secondary immunodeficiencyCirrhosisEnteritis due to radiationDiabetes mellitusPancreatic insufficiency: chronic pancreatitis, cystic fibrosis Terminal renal disease Celiac disease |
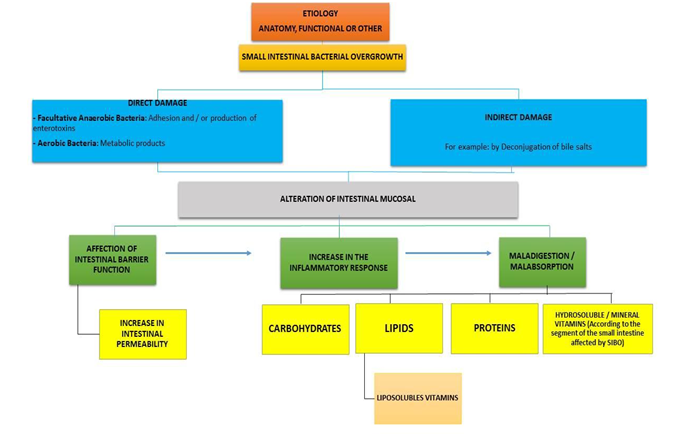
Figure 1: Consequences of SIBO[20].
Consequences of bacterial overgrowth
The impact of SIBO on the intestine can be divided into (Figure 1):
• Impact at the level of the mucus
• Alteration of the absorption of the nutrients.
Damage to the intestinal mucus – Inflammation
The increase in facultative anaerobic bacteria causes epithelial lesion due to direct adherence, which favors the production of ammonium, D-lactate, peptidoglycans, and endotoxins, and stimulates the synthesis and secretion of pro inflammatory cytokines[6,9]. Furthermore, the aerobic bacteria synthesize enzymes, and the products of their metabolism damage the mucus, such as, for example, secondary biliary acids, which result in disjoining and dehydroxylation of the primary biliary acids. They are directly erosive of the mucus of the jejunum[11,12]. These changes induce inflammation, with activation of the interleukins and the inflammatory mediators, which determine an increase in the production of the mucus that interrupts or inhibits absorption[12,13].
Malabsorption of nutrients
The alteration in the critical micellar concentration, a consequence of the disjoining and dehydroxylation of the biliary acids, and concomitantly of the alteration of the enterohepatic circle, negatively impacts the digestion and absorption of fatty acids[9]. Therefore, steatorrhea and secondary deficiency of liposoluble vitamins are favored. On the other hand, malabsorption of carbohydrates also occurs, due to the intraluminal bacterial degradation of carbohydrates, as well as to the damage of the brush border, a specific place for terminal digestion[12,13]. Due to this, the digestion and absorption of monosaccharides is altered, because lactose intolerance is common in these patients[9,12,13]. Also, secondary loss of protein enteropathies and decrease in the levels of enterokinase, which determine the effect on the digestion and the absorption of proteins, have been described[6,9].
In addition to deficiencies of liposoluble vitamins, deficiencies of hydrosoluble vitamins have been described, such as B1 (thiamin), nicotinamide, and B12. The alteration in the levels of hydrosoluble vitamins could be related to consumption by anaerobic bacteria before they are absorbed[9].
Clinical manifestations of Small Intestine Bacterial Overgrowth
The clinical signs of SIBO are ample, and include mild symptoms that can pass unnoticed. In general, the signs and symptoms are secondary to malabsorption of nutrients. Digestive and extra digestive manifestations have been described, which can be secondary to the etiology of SIBO and /or the deficiencies of certain nutrients (Table 2)[12,13].
SIBO is one of the causes of chronic abdominal pain. This can be illustrated with the findings of a study of children with abdominal pain, where it was found that 63% had positive levels for the hydrogen breath test[6].
Digestive manifestations such as abdominal pain, abdominal distension, and alternating depositions (diarrhea interspersed with periods of constipation), nonspecific and non-pathognomonic, can guide the diagnosis of Irritable Bowel Syndrome (IBS). Nevertheless, they can also mask a SIBO in the strict sense. Likewise, IBS can occur concomitantly with SIBO[14].
In patients with celiac disease, a prevalence of SIBO of 9& to 55% has been reported, being greater in patients with persistent symptomology and those who do not respond to a gluten-free diet[9,13,15].
Table 2: Digestive and extradigestive manifestations of Small Intestinal Bacterial Overgrowth (SIBO) syndrome[6,13,20].
| Digestive Manifestations | Extradigestive manifestations |
| VomitingLack of appetiteDyspeptic Symptoms Abdominal pain Abdominal Distension MeteorismPersistent and/or chronic diarrhea Perineal erythema SteatorrheaManifestations associated with deficiencies of micronutrients (vitamins A, D, E, B12, thiamin, nicotinamide, iron)Malnutrition | Dermatological (dermatitis)Neurological (peripheral neuropathies)Osteomuscular (arthralgias) |
Diagnosis of Small Intestine Bacterial Overgrowth
An urgent and detailed clinical history is the fundamental tool for the diagnostic route and for defining the adequate approach, according to the etiology responsible for SIBO. Through it, the origin should be elucidated, and the ordering of unnecessary lab tests will be avoided, which increase costs and/or lead to inadequate treatments.
As part of the history, it is worthwhile to ask about the clinical manifestations, keep in mind the time of evolution and the associated symptoms, and detect signs and/or symptoms that can suggest organic pathogens. Within this framework, the pediatrician should be attentive to some signs for alarm that can alert him with respect to SIBO (Table 3).
Table 3: Signs of alarm in the investigation of SIBO[6].
| Newborns and infants | Preschoolers, school-age, and adolescents |
| Growth deficiency | Growth deficiency |
| Hematochezia | Nocturnal and incapacitating abdominal pain |
| Chronic nocturnal diarrhea | Hematochezia |
| Nocturnal and/or projectile vomiting | Loss of weight |
| Steatorrhea | Persistent Fever |
| DysphagiaHepatomegaly | Arthralgias |
| Alterations in the state de consciousness | Oral ulcers |
| Nocturnal and/or projectile vomiting | |
| Neurological manifestations: alterations in neurodevelopment, hypotonicity, puffy fontanelle, microcephaly or macrocephaly | Chronic nocturnal diarrhea HepatomegalyDysphagia |
The clinical manifestations vary according to the age group, and also in accordance with how the motive for the consultation varies. In newborns and infants, the parents usually consult due to lack of appetite, alterations in depositions (persistent and/or chronic diarrhea, constipation), abdominal distension, meteorism, and abdominal pain. In preschoolers, school-age children and adolescents, the most common symptom is abdominal pain, as the only manifestation or associated with other digestive symptoms[9].
Likewise, it is important to ask if there were previous treatments and if acid suppressants, antibiotics or antiparasitics have been administered. Another fundamental point is to analyze the feeding of the child, checking if he consumes beverages with a high content of carbohydrates (juices and panela water, among others), as well as the volume consumed, and if he receives Infant Formula (IF), the type and quantity and if the IF has been changed during the course of the illness, and if he ingests foods that are a source of fiber, among others. Furthermore, it should be clarified if modifications in diet have been made during the course of the illness as a part of the treatment, and if so, what (such as the exclusion of lactose or saccharine and restriction of cow’s milk, among others), and whether or not this has had a satisfactory result for the improvement of the patient.
With respect to the history, it is important to gather data about the gestational age (prematurity), antibiotic therapy in the mother, and the method of birth. In the postnatal period, one should ask about the time of evacuation of the meconium (the meconial ileum can cause suspicion of cystic fibrosis), if there was maternal breastfeeding and its duration, whether it was exclusive, the age of introduction of complementary feeding, hospitalizations, and antibiotic therapy. All the above-mentioned aspects are factors that can alter the intestinal microbiota and predispose for illnesses such as constipation and food allergy, among others. Furthermore, if the patient has had a surgical procedure that has required resection of intestinal segments, it is indispensable to determine what region was resected and if the ileocecal valve was conserved, among others[13].
The physical exam should be complete, and it should be observed if there is abdominal distension, colic cord, hepatosplenomegaly, perineal erythema, or as cites, as well as extra digestive findings that suggest specific nutritional deficiencies. Furthermore, a diagnosis should be made of the nutritional state by means of anthropometric indicators, and ideally the findings should be contrasted with previous data, since lack of growth that coincides with the introduction of gluten and certain symptoms could help with the differential diagnosis such as that of celiac disease[9,13].
As far as paraclinical support, your request should be individualized according to the manifestations and what has been found clinically in each patient. In those that have chronic and/or persistent diarrhea, extra tests could be needed in order to discount etiologies such as celiac disease, cystic fibrosis, and food allergy, among others[9]. In children with chronic abdominal pain and dyspeptic symptoms, an upper gastrointestinal endoscopic exam could be required, with a biopsy.
As was previously described, in patients with SIBO there is malabsorption of nutrients and in some of them macrocytic or microcytic anemia, low levels of vitamin B12, serum prealbumin and transferring, and lymphopenia is seen. Additionally, augmented fecal a1-antitrypsin can also be found in patients with malabsorption of proteins or elevated acid steoatocrit in children with malabsorption of fats[9,13]. Focus on malabsorption syndrome requires special consideration, although it is not the objective of the present article.
The hydrogen breath test is the most common one used for diagnosing SIBO. It is a non-invasive, low-risk, economical exam that is easy to implement. A dose of carbohydrates is given orally, for example lactulose. If there is an excess of bacteria in the small intestine, the production of hydrogen increases, because carbohydrates are metabolized by the small intestine. It is considered to be positive if there is an increase in hydrogen > 20 Parts Per Million (ppm) between 90 and 120 minutes after the administration of the substrate[9,12,13]. However, some factors can alter the results, among them diet, exercise, the recent use of antibiotics, and rapid orocecal transit[9]. The hydrogen breath test is more precise than the methane breath test; it has a sensitivity of 62.5% and a specificity of 82%[13]. A test of hydrogen done with lactose, if the result is positive, can indicate lactose intolerance and/or SIBO[9].
Other non-invasive tests have been described. For example, the advantage of measuring products of bacterial metabolism in urine or blood and the breath test with marked glycolate (C14) has been evaluated. However, many studies are needed on its sensitivity and specificity[9,12,13,15]. The test of reference is the culture of intestinal aspirate test, which can be obtained by endoscopy. However, it is invasive, it is not easy to administer in all patients, and it can be contaminated at the time of the sampling. Furthermore, it is difficult to cultivate the microorganisms[9,15].
Treatment of Small Intestine Bacterial Overgrowth
The treatment should be individualized. In the first place, it is worthwhile to identify determining cause in order to treat it[16]. Nevertheless, the fundamental pillar is nutritional management with dietary modifications, according to the cause and the evolution of the patient. The majority of children need restriction of simple sugars, lactose and/or saccharine, and their source foods[16,17]. When it is merited, the deficits of specific nutrients should be corrected, such as, for example, liposoluble vitamins and iron, among others[9,16]. On the other hand, the use of prokinetics has been evaluated when there are motility disorders. However, there is a lack of studies that justify their routine use[9].
In the treatment of SIBO, the goal is not to eliminate the intestinal microbiota in order to obtain a sterile intestine, but to decrease the pathogenic bacteria and modify its composition[13]. Due to the great quantity of microorganisms present, one seeks to implement an antimicrobial therapy that covers aerobic and anaerobic bacteria[9,12,13]. Treatment with a wide-spectrum antibiotic generally is effective for alleviating the symptoms, but various cycles of antibiotics are required[16]. The duration of the antibiotic depends on the symptoms and the clinical history of the patient. Cycles of 7 to 14 days have been recommended, trying to rotate the antibiotics in order to decrease the possibility of resistance[16].
For a long time, tetracycline was the treatment of choice[13]. Rifaximin is a non-systemic semi synthetic antibiotic that acts on gram-positive and gram-negative bacteria, both aerobic and anaerobic[13,16]. According to various studies, rifamixin improves the symptoms in 33% to 92% of patients, eradicating SIBO in up to 80%[13]. Rifaximin is probably the only antibiotic capable of achieving a favorable long-term clinical effect in patients with IBS (note to the executive editor: it seems to me that the complete name should not be erased, since it is the first time this syndrome is mentioned, and so I think that the name should be mentioned first and later the abbreviation, that is to say: irritable bowel syndrome IBS) and SIBO[13,16]. This antibiotic is lightly absorbed in the gastrointestinal tract, it has scarce secondary effects, and it has a low rate of resistance[6]. Di Stafano and his collaborators found that metronidazole notably reduced the positive results of the hydrogen breath tests, as well as the symptoms of the patients, although Rifaximin turned out to be more effective than metronidazole[17].
In a systematic review and meta-analysis in which the clinical effectiveness of antibiotic therapies for the treatment of patients symptomatic of SIBO was compared, Rifaximin was the most-used antibiotic[16]. Fumari and his collaborators reported that 87% of the patients treated orally with Rifaximin showed clinical improvement, and after treatment, when the non-invasive tests were evaluated, the values suggestive of SIBO decreased[18].
The use of prebiotics and probiotics has also been postulated as adjuvants in the treatment, although the evidence is limited due to the multiple etiologies that lead to SIBO[9]. Hegar and his collaborators carried out a controlled double-blind study with a placebo in 70 children treated with omeprazole for 4 weeks, in whom Lactobacillus rhamnosus R0011 and Lactobacillus acidophilus R005 were simultaneously administered. After one month of treatment, 30% showed a positive breath test suggestive of SIBO, and 62% of that 30% continued to be symptomatic. There were no differences in the results of the tests in patients who received probiotics vs the control group[19]. Other alternative forms of management are cyclical cleaning of the small intestine, especially in cases of recurrence, or else surgical treatment in patients with anatomical alterations[9,12,13]. Likewise, it is worth it to consider an intestinal transplant in patients with short bowel syndrome[9,13]. The prognosis for SIBO principally depends on the underlying illness that favors the bacterial proliferation[9]. After a successful treatment, the rate of recurrence is high. More studies are needed on the diagnosis, approach, and follow-up of these patients[13].
Conclusion and recommendations
Small Intestinal Bacterial Overgrowth (SIBO) syndrome is an entity little known and recognized by professionals in the pediatric area. Some common digestive manifestations, frequent motives for consultations, such as abdominal pain, abdominal distension, meteorism, unimproved or persistent diarrhea, and vomiting, among others, could be associated with SIBO.
For that reason, it is important to identify patients that are admitted to the emergency room, those hospitalized, or those who arrive for an out-patient consultation who have a greater risk for SIBO. Among them, it is worthwhile to concentrate on those who have:
> Enteroenteric fistulas
> Intestinal stenosis
> Resection of the ileocecal valve
> Short bowel syndrome
> Irritable bowel syndrome
> Cystic fibrosis
> Antibiotic therapy
> Treatment with acid suppressers
> X-ray treatment
The foregoing is offered especially because of the diagnostic dilatation of SIBO, the uncertainty, and the inadequate approach, as well as the negative impact on and the consequences for the health of the patient and his or her quality of life and that of his or her family.
Conflict of interest: Authors doesn’t have any conflict of interest.
References
1. Jandhyala, S.M., Talukdar, R., Subramanyam, C., et al. Role of the normal gut microbiota. (2015) World J Gastroenterol 21(29): 8787-8803.
2. Jost, T., Lacroix, C., Braegger, C., et al. Impact of human milk bacteria and oligosaccharides on neonatal gut microbiota establishment and gut health. (2015) Nutr Rev 73(7): 426-437.
3. Pacheco, A.R., Barile, D., Underwood, M.A., et al. The impact of the milk glycobiome on the neonate guts microbiota. (2015) Annu Rev Anim Biosci 3: 419-445.
4. Devaraj, S., Hemarajata, P., Versalovic, J. La microbiota intestinal humana y el metabolismo corporal: Implicaciones con la obesidad y la diabetes. (2013) Acta Bioquim Clin Latinoam 47(2): 421-434.
Pubmed||Crossref||Others
5. Bibiloni, R., Membrez, M., Chou, C.J., et al. Microbiota intestinal, obesidad y diabetes. (2009) Ann Nestle [Esp] 67: 39–48
Pubmed||Crossref||Others
6. Siniewicz-Luze||czyk, K., Bik-Gawin, A., Zeman, K., et al. Small intestinal bacterial overgrowth syndrome in children. (2015) Prz Gastroenterol 10(1): 28-32.
7. Marques, T.M., Cryan, J.F., Shanahan, F., et al. Gut microbiota modulation and implications for host health: Dietary strategies to influence the gut–brain axis. (2014) Innovat Food Sci and Emerg Technol 22: 239–247.
8. Grace, E., Shaw, C., Whelan, K., et al. Review article: small intestinal bacterial overgrowth – prevalence, clinical features, current and developing diagnostic tests, and treatment. (2013) Aliment Pharmacol Ther 38(7): 674-688.
9. Parish, C.R. Nutritional Consequences of Small Intestinal Bacterial Overgrowth. (2008) Pract Gastroenterol 69: 15-28.
Pubmed||Crossref||Others
10. Sieczkowska, A., Landowski, P., Kaminska, B., et al. Small Bowel Bacterial Overgrowth in Children: A Comprehensive Review. (2016) J Pediatr Gastroenterol Nutr 62(2): 196-207.
11. Ierardi, E., Losurdo, G., Sorrentino, C., et al. Macronutrient intakes in obese subjects with or without small intestinal bacterial overgrowth: an alimentary survey. (2016) Scand J Gastroenterol 51(3): 277-280.
12. Sachdev, A.H., Pimentel, M. Gastrointestinal bacterial overgrowth: pathogenesis and clinical significance. (2013) Ther Adv Chronic Dis 4(5): 223-231.
13. Bures, J., Cyrany, J., Kohoutova, D., et al. Small intestinal bacterial overgrowth syndrome. (2010) World J Gastroenterol 16(24): 2978-2990.
Pubmed||Crossref||Others
14. G||siorowska, J., Czerwionka-Szaflarska, M. Small intestinal bacterial overgrowth syndrome and irritable bowel syndrome [Polish]. (2013) Prz Gastroenterol 8: 165-171.
Pubmed||Crossref||Others
15. Khoshini, R., Dai, S.C., Lezcano, S., et al. A systematic review of diagnostic tests for small intestinal bacterial overgrowth. (2008) Dig Dis Sci 53(6): 1443-1454.
16. Shah, S.C., Day, L.W., Somsouk, M., et al. Meta-analysis: antibiotic therapy for small intestinal bacterial overgrowth. (2013) Aliment Pharmacol Ther 38(8): 925-934.
17. Di Stefano, M., Malservisi, S., Veneto, G., et al. Rifaximin versus chlortetracycline in the short-term treatment of small intestinal bacterial overgrowth. (2000) Ali Pharmacol Ther 14(5): 551-556.
18. Furnari, M., Parodi, A., Gemignani, L., et al. Clinical trial: the combination of rifaximin with partially hydrolysed guar gum is more effective than rifaximin alone in eradicating small intestinal bacterial overgrowth. (2010) Aliment Pharmacol Ther 32(8): 1000-1006.
19. Hegar, B., Hutapea, E.I., Advani, N., et al. A double-blind placebo-controlled randomized trial on probiotics in small bowel bacterial overgrowth in children treated with omeprazole. (2013) J Pediatr (Rio J) 89(4): 381-387.
20. DiBaise, J.K. Nutritional consequences of small intestinal bacterial overgrowth. (2008) Pract Gastroenterol 69: 15–28.
Pubmed||Crossref||Others