Molecular Profiling of an Exceptional Responder and a Non-Responder to Platinum Based Chemotherapy in Two Patients with Advanced Urothelial Cancer
Tingrui Wang , Zonghui Ding
Affiliation
- 1Department of Internal Medicine, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
- 2Department of Biochemistry and Molecular Biology, Mayo Clinic Arizona, Scottsdale, Arizona
- 3The University of Arizona Cancer Center at St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Corresponding Author
Jue Wang, The University of Arizona Cancer Center at St. Joseph’s Hospital and Medical Center, 625 N 6th Street, Phoenix, AZ 85004, Tel: +1-602-406-8222; E-mail: jue.wang@dignityhealth.org
Citation
Wang, T., et al. Molecular Profiling of an Exceptional Responder and a Non-Responder to Platinum Based Chemotherapy in Two Patients with Advanced Urothelial Cancer. (2016) Clin Trials Case Stud 1(1): 35- 38.
Copy rights
© 2016 Wang, J. This is an Open access article distributed under the terms of Creative Commons Attribution 4.0 International License.
Keywords
Molecular profiling; Gemcitabine; Cisplatin; Urothelial cancer;
Abstract
Background: Understanding the molecular mechanisms of sensitivity to anticancer therapies may improve patient selection, response to therapy, and clinical trial designs. One approach to increase this understanding involves detailed studies of exceptional responders who achieved exquisite sensitivity or durable responses to therapy, and non- responders who have intrinsic or acquired drug resistance to therapy.
Case presentation: Case 1: A 72-year-old man developed multiple lung metastases three years after initial diagnosis of low-grade urothelial carcinoma followed with left nephroureterectomy and multiple courses of intra-vesical BCG therapy. He was enrolled into a randomized, phase III study of gemcitabine/cisplatin plus/minus bevacizumab therapy. He had a near complete response after two cycles of chemotherapy. The samples from his primary tumor and metastatic lesions were examined with a battery of immunohistochemical and molecular assays.
Case 2: A 41-year-old female with history of pT3bpN2, high-grade transitional cell carcinoma of the bladder, status post radical cystectomy received four cycles of adjuvant gemcitabine/cisplatin. Three months afterwards, she was found with large pelvic metastases. The samples from her pelvic mass were examined with immunohistochemical and molecular assays.
Conclusion: Case 1 patient has low expression of ERCC1 and RRM1 and mutations of BRCA1 and ATM. Case 2 patient has amplification of AKT1. The molecular profiling of two cases provides potential explanation for the exceptional response and non-response to gemcitabine/cisplatin respectively. Other actionable gene alterations provide rational basis for future targets. An in-depth analysis of tumor molecular profile will facilitate new drug development and provide new insights into urothelial carcinoma.
Introduction
Urothelial cancer is the fourth most common cancer in men in the U.S[1]. More than 76,960 Americans will be diagnosed with bladder cancer this year (58,950 men and 18,010 women), and more than 16,390 (11,820 men and 4,570 women) can expect to die of their disease[1]. Advances in the management of metastatic bladder cancer have been limited. Chemotherapy with platinum-based regimes remains the mainstay of first-line treatment and standard of care in metastatic urothelial carcinoma.
Antitumor effect of cisplatin chemotherapy is attributed to formation of platinum DNA adducts and other DNA helix-distorting lesions which could be repaired by the nucleotide excision repair (NER) system[2,3]. Excision repair cross complementation group 1 enzyme (ERCC1) exerts an important role in NER pathway and removal of platinum DNA adducts induced by cisplatin[4]. In bladder cancer, several studies have shown that ERCC1 is a potential prognostic and predictive marker of the efficacy of platinum based chemotherapy[5-8]. High tumor expression of ERCC1 is correlated with shorter survival in those with platinum based chemotherapy[6].
Ribonucleotide reductase subunit M1 (RRM1) is the regulatory subunit of ribonucleotide reductase (RR) which converts ribonucleotides to deoxynucleotides and participates in DNA synthesis and repair. Gemcitabine, a pyrimidine nucleoside antimetabolite, inhibits DNA synthesis by inhibiting RR. Several studies have suggested the RRM1 is associated with resistance to gemcitabine based chemotherapy[9]. Breast cancer 1 (BRCA1) plays a key role in DNA repair as well and low levels of BRCA1 can predict response to cisplatin. Decreased BRCA1 expression in bladder cancer resulted in greater cisplatin sensitivity[10]. Akt (protein kinase B) is a serine/threonine kinase which plays an important role in cellular survival. Akt is activated in response to growth factors and once activated Akt exerts anti-apoptotic effects and contributes to chemotherapeutic resistance. One study showed that Akt mediated BAD phosphorylation led to paclitaxel resistance in T24 human bladder cancer cells[11]. However, the case that Akt promotes resistance to gemcitabine/cisplatin has not been reported.
Understanding the molecular mechanisms of sensitivity to anticancer therapies and genetic basis of chemotherapy response may improve patient selection, rational treatment designs and response to therapy. One approach to increase this understanding involves detailed studies of exceptional responders and non-responders to gemcitabine/cisplatin based chemotherapy.
Case Presentation
Case 1
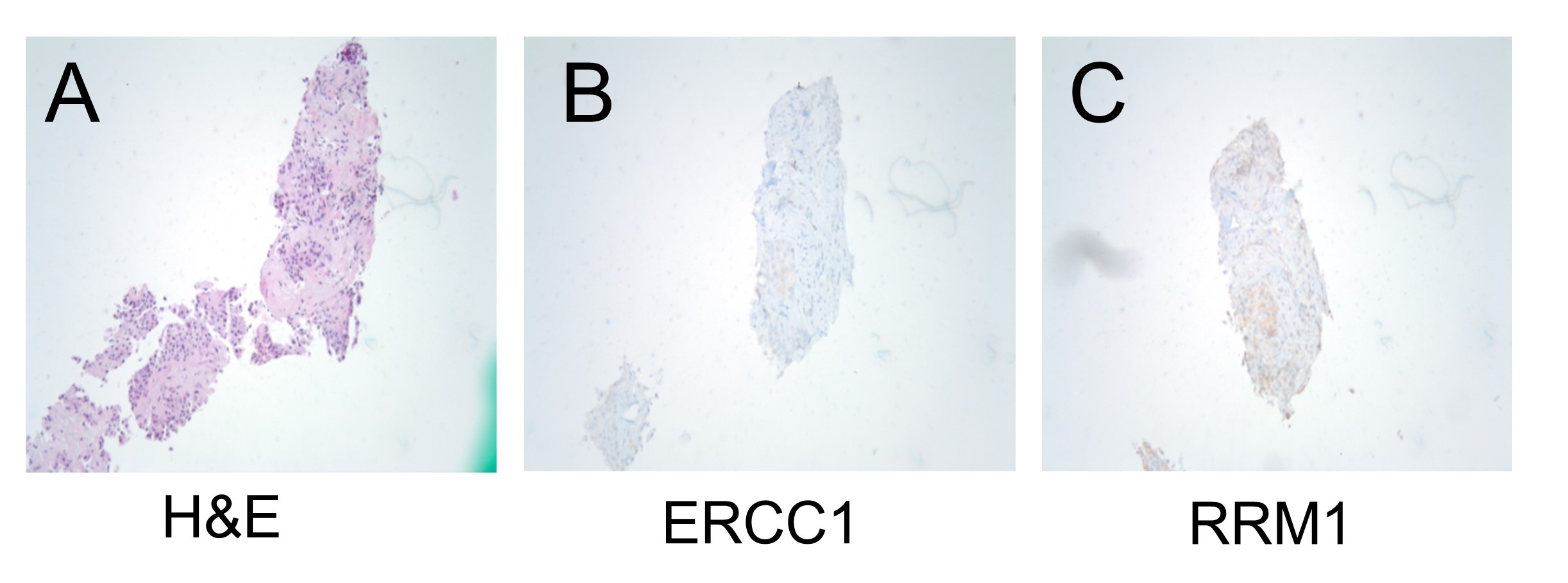
A 72-year-old man developed multiple lung metastases three years after initial diagnosis of low-grade papillary urothelial carcinoma followed with left nephroureterectomy and multiple courses of intravesical BCG therapy. One lung metastasis lesion was biopsied and pathology confirmed the diagnosis of metastatic urothelial cancer. The malignant cells were immunohistochemically positive for CK7, p63, p40, S100, and CK5/6. Stains for Napsin A and TTF1 were all negative. The tumor expressed high level of TOPO1, TLE3, MGMT, EGFR, SPARC, PGP and TUBB3, and negative for ERCC1, PTEN, TS, TOP2A, and RRM1 (Figure 1). Molecular study of his nephrectomy sample with ASHION Analytics GEM Cancer Panel™ showed multiple genomic alterations including ATM (G204); CCND1 amplification; FGFR3 (S249C); PTEN deletion. A Cell-free Tumor DNA test showed multiple alterations including FGFR3 S249C; RET E595E; TP53 V173M; TP53 C176*; TP53 V143M; BRCA1 Q780*.
Figure 1: Haematoxylin and eosin (H & E) staining (A) and IHC for ERCC1 (B) and RRM1 (C) were performed on the samples derived from lung metastases.
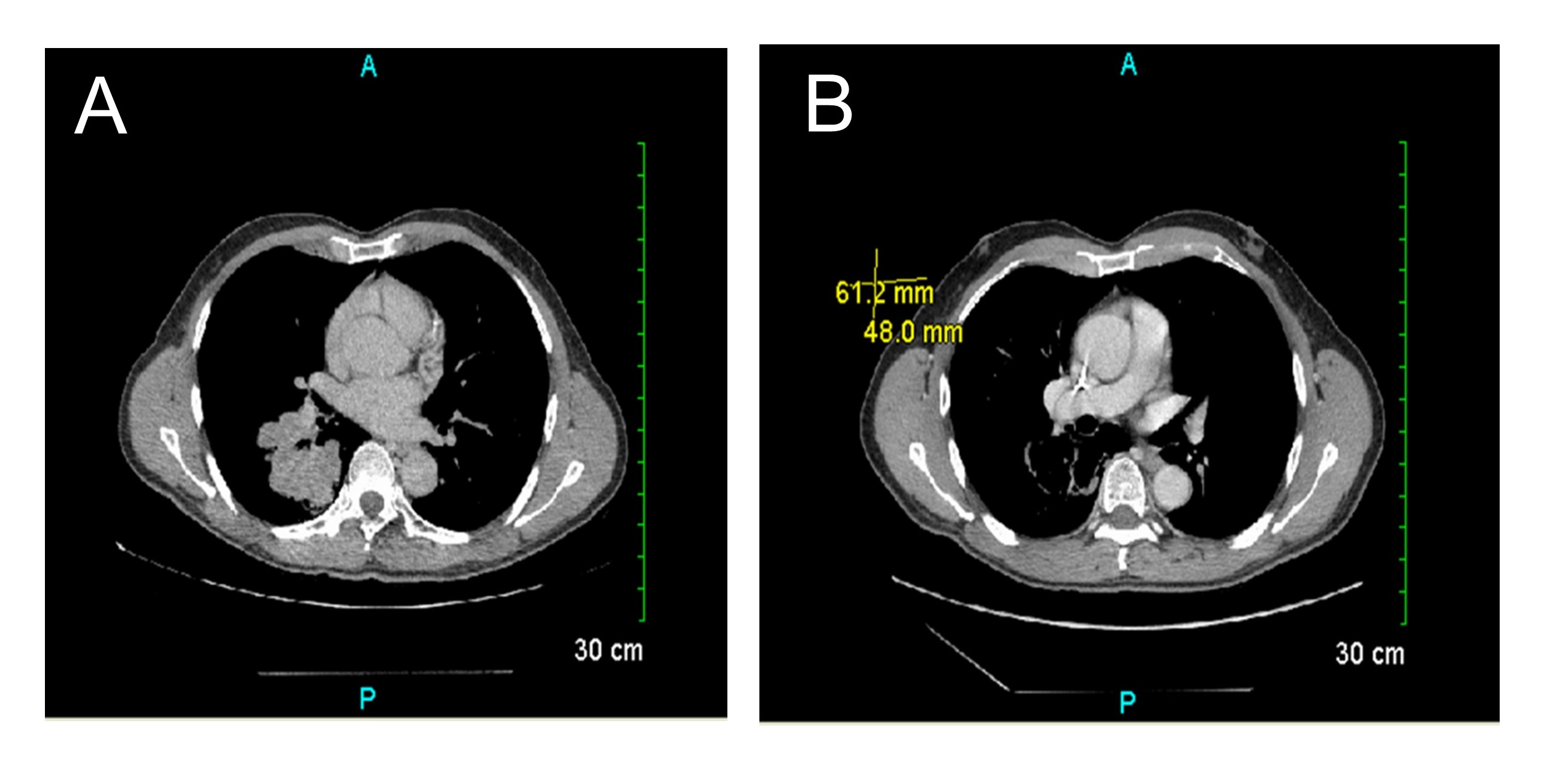
He was enrolled into a randomized, phase III study of gemcitabine/cisplatin plus/minus bevacizumab therapy for patients with metastatic urothelial cancer. He had a near complete response after two cycles of chemotherapy on the clinical trial. Chest CT was obtained before the therapy started, which showed a mass 61.2 mm x 48.0 mm in right upper lung. The lung mass remarkably decreased after two cycles (Figure 2). The patient subsequently received additional therapies at the time of disease progression and survived for more than five years.
Figure 2: A chest CT: lung before (A) and after (B) treatment of gemcitabine and cisplatin plus/minus bevacizumab therapy
Case 2
A 41-year-old female presented with pT3bpN2 highgrade transitional cell carcinoma of the urinary bladder underwent radical cystectomy followed with four cycles of gemcitabine/cisplatin after surgery. Three months afterwards, she was found with large pelvic mass 9.50 cm in greatest diameter (Figure 3) and CT guided biopsy of pelvic mass confirmed metastatic high grade urothelial carcinoma. She subsequently developed liver metastasis and died one year after initial diagnosis. The tumor was immunohistochemically strong positive for GATA3 and p63. Molecular study of her pelvic mass with ASHION Analytics GEM Cancer PanelTM showed multiple genomic alterations including AKT1 amplification, DDR2(S755Y), RAF1 amplification.
Figure 3: An Abdominal/Pelvic CT: Pelvis before (A) and growth of pelvic mass (B), greatest diameter 9.50 cm, 3 months after gemcitabine/cisplatin chemotherapy.
Discussion
Genomic profiling is a useful tool for understanding the molecular mechanism and etiology of urothelial carcinoma. Although molecular biomarkers are routinely used clinically to guide selected treatment for melanoma, lung cancer, colon cancer, the genomic molecular markers for bladder which predict the response to therapy are relatively lacking[12]. The study of extraordinary responses and non-response to chemotherapy has the potential to identify the novel molecular mechanisms of sensitivity and resistance to antitumor therapies. The low expression of ERCC1 and RRM1 and genomic alterations of BRCA1 and ATM in case 1 patient, likely explain the exceptional response to gemcitabine and cisplatin. Defective DNA repair renders tumor sensitive to cisplatin. The excision repair cross-complementation group 1 (ERCC1) is an important enzyme within the nucleotide excision repair (NER) pathway. Cisplatin induces DNA damage by formation of platinum-DNA adducts causing intra-strand and inter-strand DNA crosslinks. Cisplatin resistance is associated with removal of the adducts by NER system. Tumor cells deficient in ERCC1 impair the capacity of NER and may be more vulnerable to cisplatin[13]. Increased ERCC1 expression cause the cisplatin resistance in bladder cancer[8], cervical[14], ovarian[15], gastric[16], colon[17], non-small cell lung cancers(NSCLC)[18]. ERCC1 may represent a predictive marker for platinum based treatment in bladder cancer. Several studies have shown that high ERCC1 mRNA or protein expression is likely to cause the cisplatin resistance phenotype and correlated with poor prognosis treated with cisplatin based chemotherapy[5]. Sun et al. has shown that ERCC1 negative tumor benefit from adjuvant gemcitabine plus cisplatin chemotherapy as measured by overall survival and disease free survival[6].
Ribonucleotide reductase M1 (RRM1) is the regulatory subunit of ribonucleotide reductase holoenzyme (RR) that involves in the production of deoxyribonucleotides for de novo DNA synthesis and DNA repair. Gemcitabine is a deoxycitidine analogue, which inhibits RR activity. RRM1 is associated with resistance to gemcitabine based chemotherapy and lower/negative RRM1 expression was associated with higher response to gemcitabine-containing treatment and better prognosis[19].
The breast cancer susceptibility gene 1 (BRCA1) is involved in the process of mitosis and also plays a predominant role in the repair of DNA damage. Loss of BRCA1 functions sensitize the activity of cisplatin in breast cancer animal model[20]. Low BRCA1 expression in bladder cancer resulted in greater cisplatin sensitivity[10].
Ataxia telangiectasia mutated (ATM) has a central role in DNA repair and it is mutated in 11% of urothelial carcinoma[21]. The response to DNA repair includes recognition of damaged DNA, repair protein, transcription regulation, and activation of apoptosis[22]. Defects in ATM increase sensitivity to chemotherapy in cell lines and animal models[23]. Higher numbers of ATM alterations correlate with response to cisplatin based chemotherapy[12]. ATM-deficient tumors may predict sensitivity to DNA damage agents such as platinum based therapy, as well as poly(ADP-ribose) polymerase (PARP) inhibitors[12]. Other actionable gene alterations (FGFR3, CCND1 and RET) provide rational basis for future targets.
Case 2 patient shows AKT1 amplification and presents with resistance to gemcitabine/cisplatin chemotherapy. Akt is cellular homologue of v-akt oncogene and has three isoforms, Akt1, Akt2, Akt3. Akt plays an important role in survival when cells are exposed to DNA damage and regulate the apoptotic machinery such as BAD or caspase-9[24]. Akt activity promotes breast cancer[25], glioma[26], NSCLC[27] progression and therapeutic resistance. Szanto et al. reported T24 human bladder cancer cells resistance to paclitaxel by Bad phosphorylation resulting from activation of PI-3K-Akt pathway and inhibiting this pathway could facilitate paclitaxel therapy[11]. Our case report first suggests that Akt also potentially induces gemcitabine/cisplatin resistance. This finding warrants further investigation in mechanism.
We realize the limitation of case study, however accumulations of these unique cases add to current understanding of precision medicine, which will be further tested in prospective trials. An in-depth analysis of tumor molecular expression in exceptional responders and non-responders may facilitate new drug development for advanced urothelial carcinoma.
Conclusion
Although next generation sequencing (NGS) of “N of 1” cases have identified mechanisms of exceptional response to investigational therapies in urothelial carcinoma, as far as we know, such approaches have not been applied to exceptional non-responders. Here, we describe NGS assessment of clinical samples from both exceptional responder and non-responder. The molecular profiling of tumors in the cases reported here provides potential explanation for the exceptional response and non-response to gemcitabine and cisplatin chemotherapy. Additional research is needed to validate our observation in prospective clinical trials.
References
- 1. Siegel, R.L., Miller, K.D., Jemal, A. Cancer statistics. (2016) CA Cancer J Clin 66(1): 7–30.
- 2. Sancar, A. Mechanisms of DNA excision repair. (1994) Science 266(5193): 1954–1956.
- 3. Zamble, D.B., Mu, D., Reardon, J.T., et al. Repair of cisplatin-DNA adducts by the mammalian excision nuclease. (1996) Biochemistry 35(31): 10004–10013.
- 4. Rosell, R., Taron, M., Ariza, A., et al. Molecular predictors of response to chemotherapy in lung cancer. (2004) Semin Oncol 31(1 Suppl 1): 20–27.
- 5. Bellmunt, J., Paz-Ares, L., Cuello, M., et al. Gene expression of ERCC1 as a novel prognostic marker in advanced bladder cancer patients receiving cisplatin-based chemotherapy. (2007) Ann Oncol 18(3):522–528.
- 6. Sun, J.M., Sung, J.Y., Park, S.H., et al. ERCC1 as a biomarker for bladder cancer patients likely to benefit from adjuvant chemotherapy. (2012) BMC Cancer 12:187.
- 7. Ozcan, M.F., Dizdar, O., Dincer,, N. et al. Low ERCC1 expression is associated with prolonged survival in patients with bladder cancer receiving platinum-based neoadjuvant chemotherapy. (2013) Urol Oncol 31(8): 1709–1715.
- 8. Kim, K.H., Do, I.G., Kim, H.S., et al. Excision repair cross-complementation group 1 (ERCC1) expression in advanced urothelial carcinoma patients receiving cisplatin-based chemotherapy. (2010) APMIS 118(12): 941–948.
- 9. Bergman, A.M., Eijk, P.P., Ruiz, V., et al. In vivo induction of resistance to gemcitabine results in increased expression of ribonucleotide reductase subunit M1 as the major determinant. (2005) Cancer 65(20): 9510–9516.
- 10. Font, A., Taron, M., Gago, J.L., et al. BRCA1 mRNA expression and outcome to neoadjuvant cisplatin-based chemotherapy in bladder cancer. (2011) Ann Oncol 26(1): 139–144.
- 11. Szanto, A., Bognar, Z., Szigeti, A., et al. Critical role of bad phosphorylation by akt in cytostatic resistance of human bladder cancer cells. (2009) Anticancer Res 29(1): 159–164.
- 12. Plimack, E.R., Dunbrack, R.L., Brennan, T.A., et al. Defects in DNA Repair Genes Predict Response to Neoadjuvant Cisplatin-based Chemotherapy in Muscle-invasive Bladder Cancer. (2015) Eur Urol 68(6): 959–967.
- 13. Li, Q., Yu, J.J., Mu, C., et al. Association between the level of ERCC-1 expression and the repair of cisplatin-induced DNA damage in human ovarian cancer cells. (2000) Anticancer Res 20(2 A): 645–652.
- 14. Britten, R., Liu, D., Tessier, A., et al. ERCC1 expression as a molecular marker of cisplatin resistance in human cervical tumor cells. (2000) Int J Cancer 89(5): 453–457.
- 15. Dabholkar, M., Vionnet, J., Bostick-Bruton, F., et al. Messenger RNA levels of XPAC and ERCC1 in ovarian cancer tissue correlate with response to platinum-based chemotherapy. (1994) J Clin Invest 94(2): 703–708.
- 16. Metzger, R., Leichman, C.G., Danenberg, K.D., et al. ERCC1 mRNA levels complement thymidylate synthase mRNA levels in predicting response and survival for gastric cancer patients receiving combination cisplatin and fluorouracil chemotherapy. (1998) J Clin Oncol 16(1): 309–316.
- 17. Shirota, Y., Stoehlmacher, J., Brabender, J., et al. ERCC1 and thymidylate synthase mRNA levels predict survival for colorectal cancer patients receiving combination oxaliplatin and fluorouracil chemotherapy. (2001) J Clin Oncol 19(23): 4298–4304
- 18. Olaussen, K.A., Dunant, A., Fouret, P., et al. DNA repair by ERCC1 in non-small-cell lung cancer and cisplatin-based adjuvant chemotherapy. (2006) N Engl J Med 355(10): 983–991.
- 19. Gong, W., Zhang, X., Wu, J., Chen, L., et al. RRM1 expression and clinical outcome of gemcitabine-containing chemotherapy for advanced non-small-cell lung cancer A meta-analysis. (2012) Lung Cancer 75(3): 374–380.
- 20. Tassone, P., Di Martino, M.T., Ventura, M., et al. Loss of BRCA1 function increases the antitumor activity of cisplatin against human breast cancer xenografts in vivo. (2009) Cancer Biol Ther 8(7): 648–653.
- 21. Cyriac, Kandoth., Michael ,D., McLellan, Fabio Vandin., et al. Mutational landscape and significance across 12 major cancer types. (2013) Nature 502(7471): 333–339.
- 22. Ahmed, M., Rahman, N. ATM and breast cancer susceptibility. (2006) Oncogene 25(43): 5906–5911.
- 23. Köcher, S., Spies, Naumann A., Kriegs, M. ATM is required for the repair of Topotecan-induced replication-associated double-strand breaks. (2013) Radiother Oncol 108(3): 409–414.
- 24. Cardone, M.H. Regulation of Cell Death Protease Caspase-9 by Phosphorylation. (1998) Science 282(5392): 1318–1321.
- 25. Clark, A.S., West, K., Streicher, S., et al. . Constitutive and inducible Akt activity promotes resistance to chemotherapy, trastuzumab, or tamoxifen in breast cancer cells. (2002) Mol Cancer Ther 1(9): 707–717.
- 26. Turner, K.M., Sun, Y., Ji, P., et al. Genomically amplified Akt3 activates DNA repair pathway and promotes glioma progression. (2015) Proc Natl Acad Sci USA 112(11): 3421–3426.
- 27. Brognard, J., Clark, A., Ni ,Y., et al. Akt/protein kinase B is constitutively active in non-small cell lung cancer cells and promotes cellular survival and resistance to chemotherapy and radiation. (2001) Cancer Res 61(10): 3986–3997.