Prenatal Intraamniotic Surfactant Administration Improves Lung Function at Birth and the Effects of Conventional Postdelivery Treatment in Animal Model
Claudio Solana3, Guillermo Lobenstein2, MatÃas Uranga Imaz2, Diego Häbich2
Affiliation
- 1Department of Obstetrics, Buenos Aires University, Fellow ACOG Buenos Aires Argentine Section, Chief of Obstetrics Service, Hospital Alemán, Buenos Aires, Argentina
- 2Department of Obstetrics and Pediatrics, Hospital Alemán, Buenos Aires, Argentina
- 3Department of Neonatology, Hospital Materno - Infantil “Dr. R. Sardá,” Buenos Aires, Argentina, Neonatal Intensive Care Unit, Hospital Alemán, Buenos Aires, Argentina
Corresponding Author
Ricardo Illia, MD, Department of Obstetrics and Pediatrics, Buenos Aires University, Fellow ACOG Buenos Aires Argentine Section, Chief of Obstetrics Service, Hospital Alemán, Avenida Pueyrredón - 1640, Buenos Aires - 1118, Argentina, Tel: +54 11 4827-7000; Fax: +54 11 4827-7000; E-mail: rhillia@gmail.com
Citation
Illia, R., et al. Prenatal Intra amniotic Surfactant Administration Improves Lung Function at Birth and the Effects of Conventional Post delivery Treatment in Animal Model: A Randomized Controlled Trial. (2016) J Gynecol Neonatal Biol 2(2): 20-23.
Copy rights
© 2016 Illia, R. This is an Open access article distributed under the terms of Creative Commons Attribution 4.0 International License.
Keywords
Amniotic fluid; In-utero therapy; preterm delivery; surfactant prophylaxis
Abstract
Introduction: Surfactant diluted in the pulmonary fluid at birth promove airway formation and prevents lung injury, for this reason some investigators have proposed that actual surfactant prophylaxis in high risk preterm deliveries, should be done before the first breath, perhaps in-uteri.
Objective: To evaluate if intra amniotic surfactant plus conventional neonatal instillation improves respiratory outcome compared with only conventional neonatal instillation.
Materials and Methods: Randomized, controlled and double blinded protocol in a model of very immature born lambs. 125 days pregnant ewes received, with ultrasound guide, an intra amniotic injection of 240 mg of natural bovine surfactant or normal saline solution. After one hour of fetal breathing movement’s stimulation with aminophylline the premature lambs fetuses were delivered by C-section, and a 3-mm endotracheal tube was placed by tracheotomy. Heart rate, aortic blood pressure, central temperature, respiratory rate and hemoglobin saturation, as well as mechanical ventilator settings, were continuously monitored. Variables under study were: Peak Inspiratory Pressure (PIP), Mean Airway Pressure (MAP), Oxygenation Index (OI) and Arterio-Alveolar Ratio (a/A).
Conclusion: Prenatal intra amniotic surfactant instillation improves short-term respiratory outcome compared with conventional postpartum treatment in this animal model.
Introduction
Antenatal glucocorticoids for fetal lung maturation[1] and postnatal prophylactic surfactant administration[2-8] are the only two proven therapies for Respiratory Distress Syndrome (RDS) prevention in premature newborn babies.
Surfactant diluted in the pulmonary fluid at birth facilitates airway formation and prevents lung injury, for this reason some investigators have proposed that actual surfactant prophylaxis in high risk preterm deliveries, should be done before the first breath, perhaps in-utero, before birth.
Under this hypothesis, Galan and colleagues in a rabbit model in 1992[9] and then in premature baboons in 1993[10], found that lungs of intra amniotically surfactant treated fetuses had improved postpartum lung mechanics and less histological lesions by microscopy. In 1997, Cosmi et al[11] published a non-controlled experience in human fetuses instilled by amniocentesis with natural surfactant and subjected to fetal breathing movements induced by endovenous aminophylline injected to the mother, with good postpartum outcome and without collateral effects. Petrikovsky et al.[12] had previously reported the feasibility of intrauterine injection of surfactant directly in the mouth of three human fetuses with a fiber optic endoscope introduced through the cervix after spontaneous rupture of the membranes without adverse effects.
The concern about this procedure is what proportion of intra amniotically given surfactant actually reaches the fetal pulmonary compartment. In a recent study[13], we found that 1 to 5% of the total dose was deposited in the lungs of guinea pigs fetuses. For this and perhaps other reasons, prenatal surfactant administration may be less effective than postnatal treatment, as Galan and colleagues reported in 1996[14] when both strategies were compared in premature baboons.
Is evident that intra amniotic surfactant administration does not replace conventional postpartum treatment, but, as far as we know, nobody has studied if both treatments, antenatal surfactant instillation into the amniotic fluid plus intratracheal postnatal treatment, improves respiratory outcome compared with only conventional neonatal instillation. With this purpose we designed this randomized, controlled and blinded protocol in a model of very immature born lambs. If our hypothesis is confirmed, this protocol could be useful in cases of very preterm labor in which ones there is no time to administrate prenatal steroids.
Materials and Methods
A 125 days pregnant ewes (equivalent to preterm human newborns at 30 gestational weeks) received with ultrasound guide, an intra amniotic injection of 240 mg of natural bovine surfactant (Baby Fact B®, Lab. Ge Me Pe, Argentina), or normal saline as placebo by random and blinded to the lambs care givers under sonographic control. The ewes were trough induced hormonal ovulation to achieve the pregnancy by veterinarian techniques (mainly with intravaginal progesterone sponges) respecting all the ethical issues in regard to research with animals. Both solutions showed us as the same picture, so, it was impossible to realize which solution received each ewe except to the director of the study who was at the same time blinded to the lamb’s treatment. The neonatologist was blinded about which lamb received surfactant or saline solution and there were no differences in the aspect of both solutions to allow them suspect what solution had received every lamb. After one hour of the last administration of Aminophylline to stimulate fetal breathing movements (total: 3 doses every hour previous to C Section as descripted by Dr. Emeraldo Cosmi -11), by intravenous injection to the ewes, the premature lambs fetuses were delivered by C-section under general anesthesia with ketamine as suggested by veterinarians. Before the extraction of the body and umbilical cord section, the lambs were partially extracted and a 3-mm endotracheal tube was placed by tracheostomy with local anesthesia. After this, the lambs were completely delivered, dried, placed under a radiant warmer, connected to a mechanical ventilator and umbilical central catheters inserted into aorta and cava vein for blood sampling, invasive blood pressure and fluids administration. Heart rate, aortic blood pressure, central temperature, respiratory rate and hemoglobin saturation, as well as mechanical ventilator settings, were continuously monitored (Hewlett-Packard® multi parametric monitors).
Initial ventilator settings were: Peak Inspiratory Pressure (PIP) 30 cm H2O, Peep 5 cm H2O, RF 60 cpm, IT 0.4 sec and FiO2 of 1. Thereafter, these settings were modified according to arterial blood gases in order to maintain pO2 and pCO2 between 45 and 55 mmHg.
Thirty minutes after birth all the animals received 100 mg/kg of the same natural surfactant by intratracheal instillation. That was considered time 0, then the animals were ventilated during two hours before being sacrificed by IV ketamine injection.
Variables under study were: Peak Inspiratory Pressure (PIP), Mean Airway Pressure (MAP), Oxygenation Index (OI) and Arterio-Alveolar Ratio (a/A) before and at 30, 60, 90 and 120 minutes after postnatal surfactant treatment and compared between groups.
Experiments were performed according to the NIH Guidance for Care and Use of Laboratory Animals and to the approved protocol passed by the Ethical and Teaching and Research Boards of the Hospital Aleman , Buenos Aires, Argentina.
Serum fetal glucose levels and hydration were maintained by continuous infusion of 100 ml/kg/hour of IV D10% and in case of arterial hypotension normal saline boluses of 20 ml/kg were injected intravenously.
Results
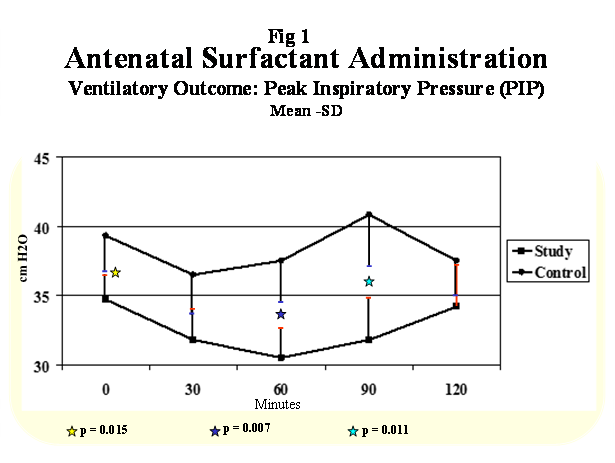
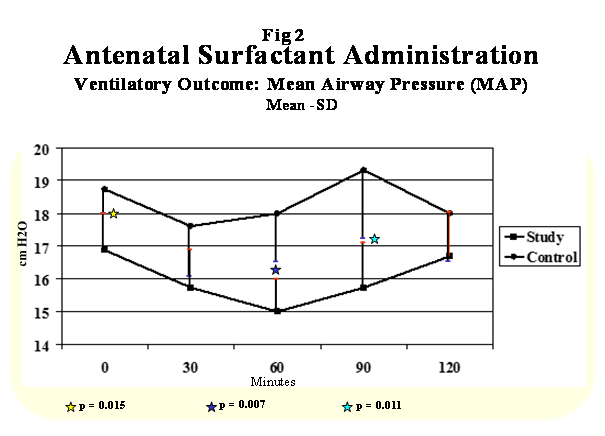
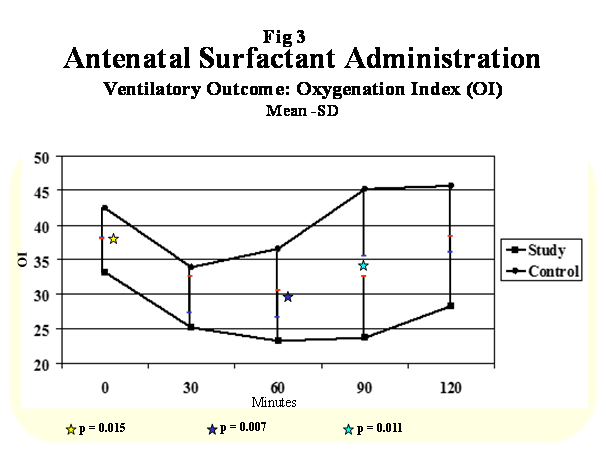
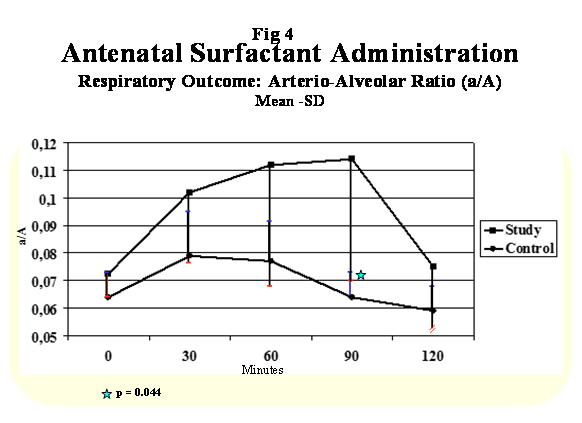
12 animals were studied, 6 in each arm. Birth weight, sex, and basal vital signs values were not different between groups at study entry. Of the main endpoints: PIP, MAP and OI were statistically lower at 30 minutes after birth, before postnatal surfactant instillation (time point 0) in antenatally treated animals and at 60 and 90 minutes after treatment, while a/A was different at time 0 and at 90 minutes only. The 120 minutes post treatment comparisons were not possible because the demise of four animals in the control group vs. only 1 in the treated group. In Figures 1 - Figure 4 PIP, MAP, IO and a/A are represented for each group in function of time as mean and SD.
Figure 1: Ventilatory Outcome: Peak Inspiratory Pressure (PIP).
Figure 2: Ventilatory Outcome: Mena Airway Pressure(MAP).
Figure 3: Ventilatory Outcome: Oxygenation Index(OI).
Figure 4: Respiratory Outcome: Arterio-Alveolar Ratio (a/A).
Discussion
This protocol was designed with the only purpose of studying if antenatal surfactant instillation of one dose of 240 mg of natural surfactant, one hour before birth improved immediate pulmonary outcome, before and after early (30 min) postnatal surfactant treatment in an animal model of very immature lungs.
The hypothesis of this trial was that if the surfactant injected into the amniotic fluid, although in a small proportional, could reach the lungs of the fetuses, all of it should be diluted in the pulmonary fluid, the exact place was it is needed in the first minutes after birth, when the airways are being formed. If this happened, initial work of breathing, need of mechanical ventilation and risks of acute lung injury could be probably reduced from birth and the results of posterior surfactant instillation improved.
In a previous study[13], we found that Tc99m labeled surfactant injected into the amniotic fluid of guinea pigs fetuses produced a lung uptake of 1 to 5% of the total administered dose, one hour later. If this could happen in this and other mammalian models, the amount of surfactant diluted in the pulmonary fluid may be similar to that present in the epithelial alveolar liquid hypo phase of term human babies (5 to 10 mg/kg).
All the animals antenatally treated with surfactant had better lung mechanics in the first 30 minutes after birth, both groups improved significantly after postnatal intratracheal surfactant instillation, being not statistically different for one hour after treatment when again in-uterus instilled group again remained with a better lung status than control animals.
Prenatal intra amniotic surfactant instillation improves short-term respiratory outcome compared with conventional postpartum treatment. We speculate that these results should be confirmed by a larger and more complex study in animals before attempting a clinical trial in humans’ beings with very preterm labor when there is no time enough to administrate prenatal steroids before delivery.
Abbreviation: PIP: Peak Inspiratory Pressure; MAP: Mean Airway Pressure; OI: Oxygenation Index; (a/A): Arterio-Alveolar Ratio
References
- 1. Crowley, P. Prophylactic corticosteroids for preterm birth. (2000) Cochrane Database Syst Rev 2: CD000065.
- 2. Horbar, J.D., Wright, E.C., Onstad, L. Decreasing mortality associated with the introduction of surfactant therapy: an observational study of neonates weighing 601 to 1300 grams at birth. The Members of the National Institute of Child Health and Human Development Neonatal Research Network. (1993) Pediatrics 92(2): 191-196.
- 3. Kendig, J.W., Notter, R.H., Cox, C., et al. A comparison of surfactant as immediate prophylaxis and as rescue therapy in newborns of less than 30 weeks’ gestation. (1991) N Engl J Med 324(13): 865-871.
- 4. Egberts, J., De Winter, J.P., Sedin, G., et al. Comparison of prophylaxis and rescue treatment with Curosurf in neonates less than 30 weeks’ gestation: a randomized trial. (1993) Pediatrics 92(6): 768-774.
- 5. Kattwinkel, J., Bloom, B., Delmore, P., et al. Prophylactic administration of calf lung surfactant extract is more effective than early treatment of respiratory distress syndrome in neonates of 29 through 32 weeks’ gestation. (1993) Pediatrics 92(1): 90-98.
- 6. Dunn, M.S., Shennan , A.T., Zayack, D., et al. Bovine surfactant replacement therapy in neonates of less than 30 weeks’ gestation: a randomized controlled trial of prophylaxis versus treatment. (1991) Pediatrics 87(3): 377-386.
- 7. Soll, R.F., Morley, C.J. Prophylactic versus selective use of surfactant for preventing morbidity and mortality in preterm infants (Cochrane review). (2012) The Cochrane Library 4.
- 8. Soll, R. Surfactant treatment of the very preterm infant. (1998) Biol Neonate 74(suppl 1): 35-42.
- 9. Galan, H.L., Kuehl, T.J. Effect of intra-amniotic administration of Exosurf in preterm rabbit fetuses. (1992) Obstet Gynecol 80(4): 604-608.
- 10. Galan, H.L., Cipriani, C., Coalson, J.J., et al. Surfactant replacement therapy in utero for prevention of hyaline membrane disease in the preterm baboon. (1993) Am J Obstet Gynecol 169(4): 817-824.
- 11. Cosmi, E.V., La Torre, R., Piazze, J.J., et al. Intra amniotic surfactant for prevention of neonatal respiratory distress syndrome (IRDS): rationale and personal experience. (1997) Eur J Obstet Gynecol Reprod Biol 71(2): 135-139.
- 12. Petrikovsky, B.M., Lysikiewicz , A., Markin, L.B., et al. In utero surfactant administration to preterm human fetuses using endoscopy. (1995) Fetal Diagn Ther 10(2): 127-130.
- 13. 13. Guinea pigs (in Press) Put Name and Magazine.
- 14. Galan, H.L., Cipriani, C., Coalson, J.J., et al. Hyaline membrane disease surfactant prophylaxis in the preterm baboon: a comparison of postpartum versus in utero therapy. (1996) Prenatal Neonatal Med 1(2): 122-130.