The Effect of Potato Polyphenols in Combination with Cellulose or Inulin in a Mixed Culture of Swine Fecal Bacteria
Aldrine Kilua1, Kyu-Ho Han1, Kenichiro Shimada1, Naoto Hashimoto2 and Michihiro Fukushima1*
Affiliation
1Department of Life and Food Sciences, Obihiro University of Agriculture and Veterinary Medicine, Inada-cho, Obihiro, Hokkaido 080-8555, Japan
2Upland Farming Resource Research Division, NARO Hokkaido Agricultural Research Center, Minami 9-4, Shinsei, Memuro, Kasai, 082-0071 Hokkaido, Japan
Corresponding Author
Michihiro Fukushima, Department of Life and Food Sciences, Obihiro University of Agriculture and Veterinary Medicine, Inada-cho, Obihiro, Hokkaido 080- 8555, Japan. Tel: 0155 49 5557/ Fax: 0155 49 5577; E-mail: fukushim@obihiro.ac.jp
Citation
Fukushima, M., et al. The Effect of Potato Polyphenols in Combination with Cellulose or Inulin in a Mixed Culture of Swine Fecal Bacteria. (2018) Int J Food Nutr Sci 5(1): 7-15.
Copy rights
© 2018 Fukushima, M. This is an Open access article distributed under the terms of Creative Commons Attribution 4.0 International License.
Keywords
Inulin; Polyphenol; In-vitro fermentation; Dietary fiber; Intestinal flora
Abstract
The effect of potato polyphenols in combination with cellulose or inulin on bacterial populations, Short Chain Fatty Acids (SCFA) production, and ammonia level during in-vitro colonic fermentation by pig colonic digests were investigated in the current study. Jar fermenters were used to conduct a small scale in-vitro colonic fermentation experiments under anaerobic condition using CO2 gas at 37°C for 48h. Four jar fermenters were assigned to one of the following four samples; Cellulose (CEL), Cellulose + Polyphenols (CP), Inulin (INU) and Inulin + Polyphenols (IP). The effect of INU or IP induced significant changes in the colonic micro biota by reducing the pathogenic bacteria as indicated by the lowering of colonic pH, and increasing the populations of beneficial bacteria compared to CEL and CP. The production of total SCFA was significantly higher in order; IP > INU > CP > CEL. Compared to INU, IP can potentially suppress bacterial populations as indicated by the reduction of butyrate production and decrease in coliform populations. While CP induced the highest level of pH and ammonia concentrations respectively, IP reduces these to significant amounts. Our results suggested that combining potato polyphenols with inulin can potentially alleviate the detrimental effects of the former during colonic fermentation and maintain a stable and healthy colonic environment.
Introduction
Dietary fibers are edible plant components or carbohydrates that are not digested and absorbed in the small intestine but fermented in the large intestine by colonic microbiota[1-3]. Epidemiological studies have shown a positive correlation between high intake of dietary fiber and health benefits. Studies have indicated that dietary fibers are instrumental in regulating body weight, immune function and contribute to diabetes control[2,4,5]. Likewise, lowering of depressive symptoms may be attributed to higher dietary fiber intake[6]. Furthermore, there is an association between adequate intake of dietary fiber and prevention of non-communicable diseases such as colon, rectal and breast cancers including coronary heart diseases[7-9].
Inulin is a soluble dietary fiber found +in various plants[10] and proven to be positively affecting the microbiota composition. Microbial response to various dietary fibers varies to some degrees and this determines the type and amount of products produced at the end of the fermentation process. With the existence of β-(2-1)-d-fructosyl fructose bonds between the fructose units along with β-configuration, the digestibility of inulin is impossible in the human small intestine[11]. Although indigestible in the small intestine, almost 90% escaped and being digested in the colon[12]. Studies confirmed and widely accepted that inulin is regarded as a prebiotic compound due to its prebiotic efficacy[13]. According to studies by Pompei et al[14] and van de Wiele et al[15], inulin selectively increases the population of bifidobacteria from proximal and distal regions of the colon while inhibiting the pathogenic ones. While inulin is fermentable in the large intestine, cellulose leaves the colon without being altered. This could be the reason why Inulin and cellulose are different with respect to their fermentation products, with the former being influential in promoting probiotic bacteria than the latter.
The microbial ecosystem has reportedly been modified by dietary polyphenols. Polyphenols are bioactive compounds besides non-digestible carbohydrates[16]. They are synthesized by plants as a defensive mechanism against radiation and microbial infection[17,18]. Depending upon the chemical structure of phenolic moiety and any attached chemical groups, polyphenols can either absorb in the small intestine or reaches the colon for microbial attack. Studies have indicated that specific polyphenols can modulate the colonic flora and ameliorate the negative effects less than prebiotics[19]. Consequently, it reduces the risk of chronic diseases caused by oxidative stress and indeed, has the prebiotic potential[19,20]. Studies have indicated that potato polyphenols contain anthocyanins derivatives[21] and can increase the population bifidobacteria and lactobacillus[22]. Other studies, however, highlighted that catabolism of polyphenols in combination with Fructo-Oligosaccharide (FOS) or Inulin increase the colonic pH slightly[23,24]. Further, polyphenols, in single supplement can increase the colonic pH to levels similar to negative control[24].
Given the strong evidence from scientific studies regarding the association of dietary fiber and health, however, the effect of potato polyphenols in combination with Cellulose or Inulin is less well appreciated. Hence, the aim of this study is to examine the hypothesis that the mixtures of potato polyphenols and inulin or resistant starch improve intestinal environment during in vitro colonic fermentation using pig colonic digest.
Materials and Methods
Preparation and determination of potato polyphenols
Preparations of potato polyphenols were done according to Han et al[25] with slight modifications. Briefly, 50 g of the sample was subjected to 5% acetic acid three times and left overnight at room temperature until almost all colored pigment was extracted. The suspension was filtered and centrifuged at 12600 rpm and 10°C for 30 minutes. The upper layers were applied directly to Diaion HP-20 resin (Nippon Rensui Co., Tokyo, Japan). The colored pigments were retained by resin and the remaining acetic acid was filtered. 1% acetic acid was used to wash the column for any impurities like proteins and salts. Finally, elution was done by using 70% ethanol. Ethanol was removed by using rotary evaporator at 35°C and the eluates were diluted using water and lyophilized to give the wanted potato extract for analysis. The total polyphenol content of potato extract was determined according to the Folin-Ciocalteu method[26]. Briefly, 0.5 ml of diluted sample was mixed with 5 ml of NaCO3 and then shaken. After 5 mins, 0.5 ml of Folin-Ciocalteu reagent was added to the mixture and vigorously shaken. After 30 mins, the absorbance was measured at 750 nm using Shimadzu 1600-UV spectrophotometer. The total phenolic content of potato extract was converted into mg gallic acid equivalents per 100 g of sample weight.
Faeces and in vitro fermentation
In vitro fermentation was conducted with a mixture of fresh Pig feces collected from a farm five minutes’ drive from Obihiro University of Agriculture and Veterinary Medicine. The pig’s feces were collected directly from the anus of three pigs and inserted in a plastic containing Anaero Pack-Anaero (Mitsubishi Gas Chemical, Tokyo, Japan). The pig feces were collected on site on the day of the experiment. Fecal slurry (10x dilution) was prepared by homogenizing equal amounts from three pigs in a stomacher (Exnizer 400, Organo Co., Tokyo, Japan) and filtered through a stomacher bag (Eiken Chemical Co., Ltd., Tochiki, Japan) filled with 0.85% NaCl solution and used as inoculums. Four pH-controlled jar fermenters (220 ml working volume, Able & Biott, Tokyo, Japan) were assigned to one of the following samples; 3% Cellulose (CEL), % Cellulose + % Potato Polyphenols (CP), 3% Inulin (INU) and % Inulin + % Potato Polyphenols (IP). The autoclaved basal nutrient broth (Difco, Sparks, MD) was added to the fermenters to give 0.8% (w/v) as a final concentration. Finally, the fermenters were inoculated with the fecal slurry to give a final concentration of 2.0% (v/v). The fermentation design was anaerobically maintained at 37°C for 48h at lower pH limit of 5.50. At 0, 6, 12, 24 and 48 h time points, pH were recorded and aliquots were collected in 2 ml tubes and stored at -30°C for analysis.
Bacteria analysis
Bacterial populations were analyzed using selective media by plate count method. Coliform bacteria and Cl. perfringens from the fermenters were inoculated and grown for one day on EMB agar (Eiken Chemical Co., Ltd.) and OPSP agar (Oxoid, Hampshire, UK) respectively. Anaerobes, Lactobacillus and Lactic Acid Bacteria (LAB) were grown for two days on BL (Eiken Chemical Co., Ltd.), Rogosa (Oxoid) and MRS (Oxoid, Hampshire, UK) agar respectively. Bifidobacterium was grown for three days on TOS-propionate agar (Yokult pharmaceutical Industry, Tokyo, Japan). Bacteria were incubated at 37°C for 24, 48 and 72h using Anaero Pack-Anaero (Mitsubishi Gas Chemical) in a sealed anaerobic container.
SCFA analysis
Aliquots from fermenters were Centrifuge at 10000 rpm and 4°C for 15 minutes. The supernatants were filtered with a 1ml syringe before 0.5N HCLO4 was added to deproteinized the filtrates. The filtrates were subjected to HPLC in a Shimadzu LC-10AD (Kyoto, Japan) equipped with a Bromothymol Blue (BTB) post-column. The conditions for analysis were done according to Han et al[27] with slight modifications.
Ammonia analysis
Aliquots from the fermenters were used to determine the ammonia nitrogen concentrations using a commercially available kit (Wako Pure Chemical Industry, Ltd., Tokyo, Japan) according to the manufacturer’s instructions.
Statistical analysis
All data are presented as a mean and Standard Error (SE). The in vitro fermentation was conducted in a block design with replicates of five. Two-way ANOVA was performed to assess the effect of fiber, extract, and their combination. Differences of p < 0.05 and p < 0.1 were taken to be statistically significant and tended to be significant respectively. If the variance was observed in the main effect of the group, Tukey’s test was used for this comparison (P < 0.05).
Results
Bacterial levels in fermenters
Table 1: Changes in bacteria populations during in vitro colonic fermentation
| CEL | CP | INU | IP | Two-Way ANOVA (p - value) | |||||
|---|---|---|---|---|---|---|---|---|---|
| Incubation time(h) | Total cell count, log10 CFU mL-1 | Fiber | Extract | Interaction | |||||
| Anaerobes | 0 | 8.16 ± 0.09 | 7.99 ± 0.05 | 8.42 ± 0.16 | 8.29 ± 0.14 | < 0.05 | 0.230 | 0.872 | |
| 6 | 8.05 ± 0.05b | 8.58 ± 0.22a | 8.80 ± 0.14a | 8.89 ± 0.09a | < 0.05 | < 0.05 | < 0.1 | ||
| 12 | 7.96 ± 0.03c | 8.53 ± 0.24b | 9.18 ± 0.14a | 9.41 ± 0.17a | < 0.05 | < 0.05 | 0.171 | ||
| 24 | 8.28 ± 0.27b | 8.52 ± 0.05b | 9.28 ± 0.20a | 9.45 ± 0.18a | < 0.01 | < 0.05 | 0.956 | ||
| 48 | 7.93 ± 0.18b | 8.43 ± 0.21ab | 8.98 ± 0.05a | 8.95 ± 0.07a | < 0.05 | < 0.1 | 0.104 | ||
| Bifidobacteria | 0 | 6.92 ± 0.13 | 6.86 ± 0.19 | 7.40 ± 0.13 | 7.26 ± 0.18 | < 0.05 | 0.370 | 0.963 | |
| 6 | 7.12 ± 0.14b | 7.72 ± 0.11a | 8.26 ± 0.20a | 8.29 ± 0.09a | < 0.05 | < 0.05 | < 0.1 | ||
| 12 | 6.94 ± 0.08c | 7.71 ± 0.15b | 8.77 ± 0.14a | 9.03 ± 0.15a | < 0.05 | < 0.05 | < 0.1 | ||
| 24 | 6.87 ± 0.14c | 7.77 ± 0.08b | 8.97 ± 0.05a | 8.98 ± 0.03a | < 0.05 | < 0.05 | < 0.05 | ||
| 48 | 6.22 ± 0.31b | 6.92 ± 0.61b | 8.85 ± 0.11a | 8.62 ± 0.20a | < 0.05 | 0.279 | < 0.05 | ||
| Lactobacilli | 0 | 6.80 ± 0.20 | 6.87 ± 0.25 | 7.22 ± 0.25 | 7.59 ± 0.14 | < 0.05 | 0.670 | 0.980 | |
| 6 | 6.85 ± 0.16 | 6.66 ± 0.28 | 7.29 ± 0.16 | 7.35 ± 0.08 | < 0.05 | 0.619 | 0.625 | ||
| 12 | 6.60 ± 0.16 | 6.69 ± 0.24 | 7.01 ± 0.22 | 7.13 ± 0.24 | < 0.05 | 0.611 | 0.976 | ||
| 24 | 6.66 ± 0.19 | 6.64 ± 0.23 | 6.81 ± 0.17 | 7.17 ± 0.20 | < 0.01 | 0.348 | 0.295 | ||
| 48 | 6.01 ± 0.32 | 6.54 ± 0.25 | 6.64 ± 0.10 | 6.49 ± 0.22 | 0.204 | 0.382 | 0.194 | ||
| Lactic acid bacteria | 0 | 6.55 ± 0.21 | 6.50 ± 0.23 | 7.12 ± 0.08 | 7.06 ± 0.10 | < 0.05 | 0.815 | 0.980 | |
| 6 | 6.63 ± 0.16 | 6.40 ± 0.21 | 6.85 ± 0.35 | 6.55 ± 0.29 | < 0.1 | 0.287 | 0.850 | ||
| 12 | 6.33 ± 0.18b | 6.39 ± 0.23b | 7.14 ± 0.13a | 6.82 ± 0.12ab | < 0.05 | 0.380 | 0.233 | ||
| 24 | 6.39 ± 0.15b | 6.43 ± 0.23b | 7.11 ± 0.15a | 7.02 ± 0.09ab | < 0.05 | 0.641 | 0.498 | ||
| 48 | 6.02 ± 0.21 | 6.32 ± 0.27 | 6.69 ± 0.21 | 6.60 ± 0.16 | < 0.05 | 0.709 | 0.348 | ||
| Coliform bacteria |
0 | 5.62 ± 0.07a | 5.56 ± 0.07a | 4.86 ± 0.10b | 4.87 ± 0.16b | < 0.05 | 0.998 | 0.954 | |
| 6 | 5.55 ± 0.07a | 5.41 ± 0.05a | 4.62 ± 0.11b | 4.78 ± 0.13b | < 0.05 | 0.909 | 0.136 | ||
| 12 | 5.45 ± 0.09a | 5.24 ± 0.15a | 4.66 ± 0.07b | 4.40 ± 0.07b | < 0.05 | 0.527 | < 0.1 | ||
| 24 | 5.57 ± 0.13a | 5.29 ± 0.11ab | 4.48 ± 0.07b | 4.59 ± 0.35c | < 0.05 | < 0.05 | 0.179 | ||
| 48 | 4.88 ± 0.16a | 4.82 ± 0.32a | 4.07 ± 0.18ab | 3.00 ± 0.37b | < 0.05 | < 0.1 | < 0.1 | ||
| Cl. Perfringes | 0 | 1.70 ± 0.31 | 1.75 ± 0.33 | 2.27 ± 0.12 | 2.25 ± 0.13 | < 0.1 | 0.883 | 0.799 | |
| 6 | 1.59 ± 0.30 | 1.57 ± 0.27 | 1.96 ± 0.14 | 1.09 ± 0.14 | 0.432 | 1.000 | 0.514 | ||
| 12 | 1.68 ± 0.29 | 1.55 ± 0.28 | 1.87 ± 0.07 | 1.72 ± 0.11 | 0.683 | 0.424 | 0.960 | ||
| 24 | 1.16 ± 0.10 | 1.16 ± 0.10 | nd | nd | 0.140 | 0.247 | 0.247 | ||
| 48 | 1.16 ± 0.10 | nd | nd | nd | 0.979 | - | - | ||
Each value represents the mean and standard error (n = 5). Two-way ANOVA was performed to assess the effect of fiber (CEL and INU), Potato polyphenols and their combination. Differences of p < 0.05 and p < 0.1 were taken to be statistically significant and tended to be significant respectively. Mean values (a - c) with unlike letters within the same point are significantly different (p < 0.05), as determined by ANOVA with Tukey’s test.nd – no detected.
[Table 1] summarizes the bacteria populations in the fermenters during in vitro colonic fermentation. The populations of bifidobacteria and anaerobic bacteria in the fermenters were significantly (p < 0.05) higher for INU and IP groups compared with CEL and CP groups respectively, although CP was significantly (p < 0.05) higher than CEL. The interaction between INU and potato polyphenols were significant (p < 0.05) for bifidobacteria at 24 and 48h. While coliform bacteria was significantly higher (p < 0.05) for CEL and CP groups, IP group was significantly (p < 0.05) lower at 24 and 48h, even lower than INU group. The populations for Lactic acid bacteria and Lactobacillus were higher for IP and INU groups compared to CP and CEL groups. There were no significant differences in the population of Cl. perfringens amongst the groups although survival can be seen only for CEL group.
SCFA concentration in fermenters
Table 2: Changes in short chain fatty acids (SCFA) during in vitro colonic fermentation.
| CEL | CP | INU | IP | Two-Way ANOVA (p - value) | |||||
|---|---|---|---|---|---|---|---|---|---|
| Incubation time (h) |
μmol ml-1 | Fiber | Extract | Interaction | |||||
| Acetate | 0 | 13.7 ± 0.6 | 16.6 ± 1.3 | 17.2 ± 1.7 | 14.7 ± 1.92 | 0.615 | 0.877 | < 0.1 | |
| 6 | 24.3 ± 1.1d | 59.9 ± 1.7b | 39.4 ± 1.6c | 78.7 ± 4.3a | < 0.05 | < 0.05 | 0.472 | ||
| 12 | 28.8 ± 1.6c | 60.3 ± 1.7b | 57.8 ± 2.6b | 129 ± 5a | < 0.05 | < 0.05 | < 0.05 | ||
| 24 | 38.0 ± 1.7d | 73.1 ± 2.3c | 95.1 ± 4.4b | 177 ± 9a | < 0.05 | < 0.05 | < 0.05 | ||
| 48 | 54.9 ± 3.4d | 100 ± 2.1c | 170 ± 7.5b | 211 ± 10a | < 0.05 | < 0.05 | 0.773 | ||
| Propionate | 0 | 3.51 ± 0.32 | 4.03 ± 0.37 | 3.96 ± 0.18 | 3.88 ± 0.29 | 0.632 | 0.476 | 0.337 | |
| 6 | 4.71 ± 0.43c | 11.1 ± 0.93b | 8.85 ± 0.46bc | 17.1 ± 1.9a | < 0.05 | < 0.05 | 0.421 | ||
| 12 | 5.48 ± 0.52b | 10.9 ± 0.9b | 12.3 ± 0.88b | 45.7 ± 6.4a | < 0.05 | < 0.05 | < 0.05 | ||
| 24 | 7.77 ± 0.54b | 13.7 ± 0.8b | 19.5 ± 1.81b | 65.8 ± 11.6a | < 0.05 | < 0.05 | < 0.05 | ||
| 48 | 13.8 ± 1.4c | 19.1 ± 1.1c | 40.7 ± 3.5b | 107 ± 6a | < 0.05 | < 0.05 | < 0.05 | ||
| Butyrate | 0 | 1.84 ± 0.15c | 2.34 ± 0.17bc | 3.20 ± 0.18a | 2.92 ± 0.15ab | < 0.05 | 0.515 | < 0.05 | |
| 6 | 2.90 ± 0.10c | 8.48 ± 0.5b | 7.96 ± 0.41b | 14.5 ± 0.7a | < 0.05 | < 0.05 | 0.328 | ||
| 12 | 3.44 ± 0.14d | 8.22 ± 0.34c | 15.5 ± 1.06b | 20.7 ± 0.65a | < 0.05 | < 0.05 | 0.741 | ||
| 24 | 4.20 ± 0.37b | 9.60 ± 0.6b | 29.3 ± 2.03a | 28.4 ± 2.4a | < 0.05 | 0.166 | < 0.1 | ||
| 48 | 6.99 ± 0.74c | 12.2 ± 0.6c | 48.5 ± 3.08a | 28.3 ± 2.4b | < 0.05 | < 0.05 | < 0.05 | ||
| Total SCFA | 0 | 19.1 ± 0.8 | 23.0 ± 1.1 | 24.3 ± 2.0 | 21.5 ± 2.0 | 0.253 | 0.728 | < 0.05 | |
| 6 | 31.9 ± 1.5d | 79.4 ± 2.6b | 56.2 ± 1.9c | 110 ± 6a | < 0.05 | < 0.05 | 0.376 | ||
| 12 | 37.7 ± 2.2c | 79.4 ± 2.7b | 85.5 ± 3.0b | 196 ± 12a | < 0.05 | < 0.05 | < 0.05 | ||
| 24 | 50.0 ± 2.4d | 96.4 ± 3.3c | 144 ± 6b | 271 ± 15a | < 0.05 | < 0.05 | < 0.05 | ||
| 48 | 75.7 ± 5.3d | 131 ± 3.3c | 259 ± 11b | 347 ± 13a | < 0.05 | < 0.05 | 0.105 | ||
Each value represents the mean and standard error (n = 5). Two-way ANOVA was performed to assess the effect of fiber (CEL and INU), Potato polyphenols and their combination. Differences of p < 0.05 and p < 0.1 were taken to be statistically significant and tended to be significant respectively. Mean values (a - c) with unlike letters within the same point are significantly different (p < 0.05), as determined by ANOVA with Tukey’s test.
[Table 2] shows the cumulative concentrations of SCFA in the fermenters during in vitro colonic fermentation. The acetic acid concentration for IP group was significantly higher (p < 0.05) than the rest of the groups at any given time points. On the other hand, CEL group was significantly (p < 0.05) lower compared with IP, INU and CP groups. It can also be noted that acetate concentration for CP group was significantly higher (p < 0.05) at 6h compared to INU group, although the concentration was significantly higher for INU group at 24 and 48h time points. The interaction between INU and potato polyphenols were significant (p < 0.05) for acetate at 12 and 24h. Propionate concentrations were significantly higher (p < 0.05) for IP group than the rest of the groups. There were no significant differences amongst other groups at 12 and 24h, although propionate concentration for INU group was significantly higher at 48h compared with CP and CEL groups. Further, the interaction between INU and potato polyphenols were significant (p < 0.05) for propionate at 12, 24 and 48h. Butyrate concentration was significantly higher (p < 0.05) at 6 and 12h time points for IP group than the rest of the groups. At 24h, there were no significant differences in the butyrate concentrations for IP and INU groups respectively. Interestingly, at 48h, the butyrate concentration for IP group was significantly (p < 0.05) lower than INU group. The interaction between INU and potato polyphenols were significant (p < 0.05) for butyrate at 48h. Likewise, significant difference (p < 0.05) for the interaction between INU and potato polyphenols can be noticed for total SCFA at 12 and 24h. The production of total SCFA were significantly higher in order; IP > INU > CP > CEL.
pH in fermenters
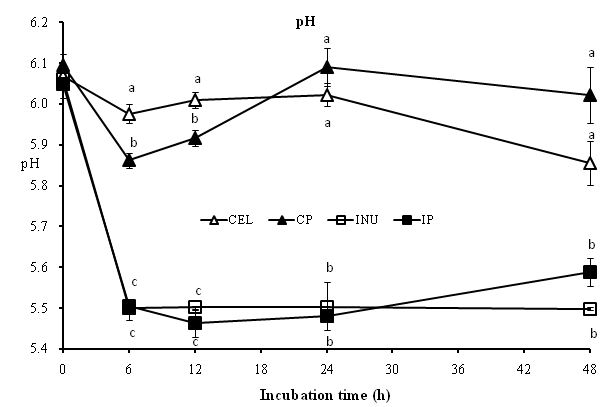
[Figure 1] shows the pH value at different time points for each sample treatments during the fermentation. The interaction between INU and potato polyphenols were significant (p < 0.05) for pH at 6h. The pH value for INU and IP groups were significantly (p < 0.05) lower compared with CEL and CP groups, although CP group was significantly (p < 0.05) lower than CEL group at 6 and 12h time points respectively. The pH values for INU and IP groups were maintained below 5.6. It can be noted at 6h that pH values make a sudden drop for all groups.
| Two-way ANOVA (P) | 0 | 6 | 12 | 24 | 48 |
|---|---|---|---|---|---|
| Fiber | 0.351 | < 0.05 | <0.05 | < 0.05 | < 0.05 |
| Extract | 0.787 | < 0.05 | < 0.05 | 0.651 | < 0.05 |
| Interaction | 0.503 | < 0.05 | 0.203 | 0.380 | 0.432 |
Figure 1: The level of pH in the fermenters during in vitro colonic fermentation at 0, 6, 12, 24 and 48h of 3% Cellulose (CEL), 3% CEL + 1 % Potato Polyphenols (CP), 3% Inulin (INU) and 3% INU + 1% Potato Polyphenols (IP). Each value represents the mean and standard error (n = 5). Two-way ANOVA was performed to assess the effect of fiber (CEL and INU), Potato polyphenols and their combination. Differences of p < 0.05 and p < 0.1 were taken to be statistically significant and tended to be significant respectively. Mean values (a - c) with unlike letters within the same point are significantly different (p < 0.05), as determined by ANOVA with Tukey’s test.
Ammonia concentrations in the fermenters
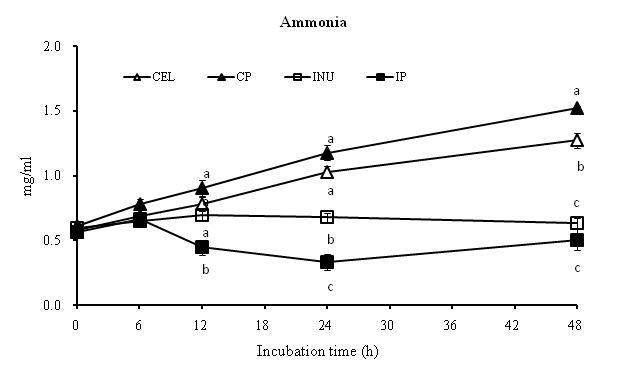
[Figure. 2] shows the ammonia concentrations of different samples treatments during in vitro colonic fermentation. The interaction between INU and potato polyphenols were significant (p < 0.05) for ammonia at 12, 24 and 48h. The ammonia concentration for IP was significantly (p < 0.05) lower than the rest of the groups at 12 and 24h time points. Although no significant difference between CEL and CP groups at 12 and 24h, at 48h, the ammonia concentration for CP group increases significantly (p < 0.05). While there was no significant difference between IP and INU groups at 48h, there was a slight decrease in ammonia concentration for IP group.
| Two-way ANOVA (P) | 0 | 6 | 12 | 24 | 48 |
|---|---|---|---|---|---|
| Fiber | 0.642 | < 0.1 | < 0.05 | < 0.05 | < 0.05 |
| Extract (Potatopolyphenol) | 0.876 | 0.168 | 0.250 | < 0.1 | 0.272 |
| Interaction | 0.372 | 0.310 | < 0.05 | < 0.05 | < 0.05 |
Figure 2: Two-way ANOVA was performed to assess the effect of fiber (CEL and INU), Potato polyphenols and their combination. Differences of p < 0.05 and p < 0.1 were taken to be statistically significant and tended to be significant respectively. Mean values (a - c) with unlike letters within the same point are significantly different (p < 0.05), as determined by ANOVA with Tukey’s test.
Discussion
The effects of potato polyphenols in combination with cellulose or inulin on colonic fermentation in a mixed culture of swine fecal bacteria were investigated in the current study. The total polyphenol content of potato extract was 6.5%, which is polyphenol-rich. A study by Han et al[25], reported the findings to be quite similar to the current result.
Studies have indicated that dietary fibers can influence or modulate the bacterial community structures of the gut[28]. Further, Russell et al[29]. Highlighted that diet is the principal factor shaping the gut bacterial composition and functionality. Consequently, Desai et al[30]. and Sonnenburg et al[31]. stated that beneficial health outcomes can be attributed to dietary fibers. Although dietary fibers may vary in structure and composition, selective consumption will determine which microbial groups are favored in the gut[32,33]. For example, inulin is a soluble dietary fiber and regarded as prebiotic[10]. Almost 90% escaped digestion and fermented in the large intestine by the colonic microbiome[34]. It was reported that upon fermentation, inulin stimulates the development of beneficial bacteria such as bifidobacteria and lactobacillus while inhibiting the pathogenic ones[35,36]. Likewise, polyphenols also stimulates beneficial bacteria by reshaping the gut microbial community and enhance beneficial effects such as weight loss[37]. Further, Parker et al[38]. reported that polyphenols can stimulate the proliferation of bifidobacteria and production of SCFA in vitro and Vendrame et al[39]. showed that fiber and polyphenols can increase bifidobacteria species.
Furthermore, a human intervention studies by Tuohy et al[40]. Reported that consuming dietary fiber and polyphenol-rich chocolate will simultaneously up regulate commensal bacteria and prevent degenerative diseases. The current study showed similar results with the population of bifidobacteria and anaerobic bacteria being higher for IP group than any other groups. When combining inulin and potato polyphenols, the populations of beneficial bacteria were increased. It is worth noticing that the interaction between INU and potato polyphenols were significant (p < 0.05) for bifidobacteria at 24 and 48h. This indicates that an increase in population was attributed to the interactions of INU and potato polyphenols. This was also supported by Parker et al[38]. In addition, Zenon et al.[24] stated that combining polyphenols and inulin increase the population of beneficial bacteria. Further, Vendrame et al.[39] showed that fiber and polyphenols can increase bifidobacteria species. Interestingly, the population of coliform bacteria was significantly (p < 0.05) lower for IP group. One possibility could be the antimicrobial effect of potato polyphenols, which needs to be further elucidated. The other possibility could be the antimicrobial effect of other species of bacteria against Coliforms. This could be the case in the current study because Francesco et al.[41] reported that Lactobacillus strains possess antimicrobial effect against gas-forming coliforms. In addition, Richard et al.[42] reported a synbiotic combination of resistant starch and bifidobacterium lactis lowered the total coli form counts. Healthy cecal environment can also be indicated by the increase of lactic acid bacteria and Lactobacillus populations. Our current result also confirmed this. In light of polyphenol consumption and CL.perfringens suppression, it was speculated previously that polyphenol consumption reduces CL.perfringens. CL.perfringens is a pathogen responsible for many gastrointestinal illnesses[43]. A study by Lopez-Legarrea et al.[44] reported that intake of polyphenol-rich fruits and vegetables increases the counts of beneficial bacteria and decreasing the population of CL.perfringens. Our current study also confirmed this. Although there was no significant difference amongst the groups, by 24h however, no CL.perfringens populations for INU and IP groups compared with CEL and CP groups.
Colonic microbiota can ferment either resistant starch or dietary fiber to SCFA at different rates and amounts, depending upon their origin[45]. An epidemiological study have also shown that SCFA (Acetate, Propionate and Butyrate) formation during colonic fermentation has been proven to exert beneficial health properties to the host[46]. Acetate, being one of the most common and abundant SCFA in human colon, metabolized in systematic areas like muscles and protect epithelial cells against pathogenic bacteria like E.coli[47-49]. According to Scheppach[50], colonic production of acetate can also enhance the ileal motility and increases colonic blood flow. Our current study showed that production of acetate was significantly (p < 0.05) higher for IP amongst the group at any given time point. The interaction between INU and potato polyphenols were significant at 12 and 24h for acetate, indicating that acetate production was influenced by the interactions of both INU and potato polyphenols. Zenon et al[24] also reported an increase in acetic acid pool when combining grapefruit flavonoids and inulin. It is worth noticing that acetic acid was the major SCFA produce followed by propionate and butyric acid. A similar result was reported by Ding et al.[51]. It was reported elsewhere that propionate also exerts some beneficial properties for the host health[51-55]. In our current study, propionate production by IP group increases significantly (p < 0.05) compared to other groups. The current result correlates well with a study by Han et al[56]. on spent turmeric powder which shows increase propionic production during colonic fermentation. The current study indicated an interaction between INU and potato polyphenols were significant at 12, 24 and 48h for propionate, highlighting the fact that propionate production was influenced by the interaction of both INU and potato polyphenols. Zenon et al[24] also reported the combination of grapefruit flavonoids and inulin increase propionate production. Health promoting effects of butyrate have also been reported elsewhere[57-60].Our current study showed increased production of butyrate during colonic fermentation except for CEL group. It is interesting to note that while CP increases butyrate production, it was suppressed by IP group, although significantly higher but lower (p < 0.05) than INU. There is a possibility that polyphenols in IP group might suppress butyrate production during colonic fermentation, although detail mechanisms need to be further elucidated. A similar finding by Zenon et al[24] also reported that combining Inulin and polyphenols significantly (p < 0.05) lowered the butyrate acid pool compared to Inulin alone. According to Negi et al[61]. Grapefruit peel extract has strong antimicrobial activity, which could possibly suppress enzymes responsible for butyrate production. In another study, Tebib et al[62] reported that grape seed tannins decreased the activity of certain bacterial enzymes. This could be the possible reason in our current study because the interaction of INU and potato polyphenols tended to be significant (p < 0.1) and significant (p < 0.05) at 24 and 48h respectively. The polyphenols in IP group might decrease the activity of bacterial enzymes responsible for butyrate production, although yet to be elucidated. Although there was a slight suppression of butyrate production, this does not affect the total SCFA production during colonic fermentation. The total SCFA production was highest (p < 0.05) for IP group in our study. In addition, the interaction of INU and potato polyphenols were significant (p < 0.05) at 12 and 24h. This is expected because an interaction of INU and potato polyphenols were significant (p < 0.05) for each SCFA.
Our current study also highlighted the effect of CP and IP during colonic fermentation. Compared to their single supplements of CEL and INU respectively, it can be noticed that pH values and ammonia concentrations were significantly (p < 0.05) higher for CP and lower (p < 0.05) for IP respectively. This could indicate potentially that combining polyphenols with dietary fiber (INU) may possibly suppress the detrimental effects exert by the former. This study corresponded well with a study by Zenon et al[24]. They reported that flavonoid extract from grapefruit used as a single dietary supplement did not display a beneficial influence on cecal metabolism, increased pH and lowered bacterial activity and SCFA concentration. Further, they stated that intake of polyphenols and Inulin simultaneously could reduce the detrimental effects of the former on cecal fermentation. This could be the case with the current study.
The colon is an active site where the microbiota normally acts upon a complex polyphenolic compounds to an absorbable molecule. The type of colonic environment will depend very much on the type and amount of substrates available in the colon. In this study, the colonic pH seems to be correlated well with the production of SCFA. It can be noted at 6h that there was a sudden drop in pH values for all treated samples. This indicates the production of SCFA during fermentation. As the production of SCFA increases, the pH value decreases. While the pH value for the positive control (INU) and IP [Figure.1] were consistent at around 5.5 (p < 0.05), CEL and CP were significantly (p < 0.05) higher. It is interesting to note that interaction of INU and potato polyphenols were significant (p < 0.05) at 6h time point. This indicates the reduction of pH is attributed by the interaction of potato polyphenols and INU because Tuohy et al.[40] highlighted that consuming dietary fiber and polyphenol-rich chocolate will simultaneously upregulate acidophilic bacteria which will ultimately reduce the colonic pH. These results may suggest that the production of different metabolites during colonic fermentation may possibly affect the colonic pH. Zenon et al[24] showed that dietary addition of inulin to the flavonoid-diet significantly decreased the pH of digest.
While lowering of colonic pH may reduce pathogenic bacteria and promote beneficial bacteria, an increase in ammonia concentration in the colon may be an important factor for colonic health as well. The colonic pH in this experiment shows the ammonia concentration during the course of fermentation of four samples. Any studies have indicated that high ammonia concentration in the colon may potentially harmful to the host. Davila et al.[63] highlighted that an increase in ammonia concentration can affect the energy metabolism of colonic epithelial cells. Further, Mitsuoka[64] stated that putrefactive compounds such as ammonia were regarded to be harmfully related to colon cancer and aging. This study showed that ammonia concentration for CP group was quite higher than the rest of the groups. Interestingly, the ammonia concentration for IP group was significantly lower (p < 0.05), even lower than control group (INU). This could indicate that addition of potato polyphenols with Inulin may suppress the formation of ammonia-producing bacteria while increasing the beneficial bacteria by lowering of colonic pH. A study conducted by Vanhoof and De Schrijver[65] reported that when inulin added to a non-purified diet on nitrogen metabolism in rats and ileum-fistulated pigs, cecal ammonia concentration decreases. In addition, Tebib et al.[62] reported that bacterial enzyme activities decrease when treated with grape seed tannins. Our current study also confirmed this because an interaction of INU and potato polyphenols were significant (p < 0.05) at 12, 24 and 48h. This could be the possible reason for the current study. While polyphenols suppress bacterial enzyme responsible for ammonia production, inulin can simultaneously reduce the colonic pH, thus, reducing the chances for pathogenic bacteria survival and maintaining a stable and healthy colonic environment simultaneously.
In conclusion, the effect of INU and IP induced significant changes in the colonic micro biota, SCFA concentrations and ammonia concentrations by reducing the colonic pH and increasing the populations of beneficial bacteria compared to CEL and CP. Therefore, our results suggested that combining potato polyphenols with Inulin can alleviate the detrimental effects of the former during colonic fermentation and maintaining a conducive environment for colonic health. Although our study highlighted the positive impact IP might have on colonic microflora, however, it is difficult to conclude with human settings because of the use of pig feces and not human feces. Further research is needed.
Conclusion
In our study, the effect of INU and IP induced significant changes in the colonic micro biota by reducing the colonic pH and increasing the populations of beneficial bacteria compared to CEL and CP. After 48h of fermentation, the production of total SCFA was significantly higher in order; IP > INU > CP > CEL. Compared to INU, IP can potentially suppress bacteria as indicated by the decrease in butyrate production and coliform populations. While the pH value and ammonia concentrations for CP increase, IP reduces these to a significant amount. Therefore, our results suggested that combining potato polyphenols with Inulin can alleviate the detrimental effects of the former during colonic fermentation and maintaining a conducive environment for colonic health.
Acknowledgement: The authors wish to sincerely thank the farm personal for providing a great support for pig fecal materials.
Funding: This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Disclosure Statement: No potential conflict of interest was reported by the authors.
References
[1] Fuller, S., Beck, E., Salman, H., et al. New horizons for the study of dietary fiber and health: a review. (2016) Plant Foods Hum Nutr 71(1): 1–12.
[2] Galisteo, M., Duarte, J., Zarzuelo, A. Effects of dietary fibers on disturbances clustered in the metabolic syndrome. (2008) J Nutr Biochem 19(2): 71– 84.
[3] British Nutrition Foundation. (2017) accessed.
Pubmed||Crossref||Others
[4] Suares, N.C., Ford, A.C. Systematic review: The effects of fiber in the management of chronic idiopathic constipation. (2011) Aliment Pharmacol Ther 33(8): 895–901.
[5]Jenkins, D.J.A., Srichaikul, K., Kendall, C.W.C., et al. The relation of low glycemic index fruit consumption to glycaemic control and risk factors for coronary heart disease in type 2 diabetes. (2010) Diabetologia 54(2): 271–279.
[6] Miki, T., Eguchi, M., Kurotani, K. Dietary fiber intake and depressive symptoms in Japanese employees: The Furukawa Nutrition and Health Study. (2016) Nutrition 32(5): 584–589.
[7] Hansen, L., Skeie, G., Landberg, R., et al. Intake of dietary fiber, especially from cereal foods, is associated with lower incidence of colon cancer in the HELGA cohort. (2011) Int J Cancer 131(2): 469–478.
[8] Holmes, M.D., Chen, W.Y., Hankinson, S.E., et al. Physical activity’s impact on the association of fat and fiber intake with survival after breast cancer. (2009) Am J Epidemiol 170(10): 1250–1256.
[9] Pereira, M.A., O’Reilly, E., Augustsson, K., et al. Dietary fiber and risk of coronary heart disease: A pooled analysis of cohort studies. (2004) Arch Intern Med164(4): 370–376.
[10] Wichienchot, S., Thammarutwasik, P., Jongjareonrak, A., et al. Extraction and analysis of prebiotics from selected plants from southern Thailand. (2011) J Sci Technol 33(5): 517–523.
Pubmed||Crossref||Others
[11] Apolinario, A.C., de Lima Damasceno, B.P., de MacedoBeltrao, N.E., et al. Inulin-type fructans: a review on different aspects of biochemical and pharmaceutical technology. (2014) Carbohydr Polym 101: 368–378.
[12] Cherbut, C. Inulin and oligofructose in the dietary fibre concept. (2002) Br J Nutr 87: 159–162.
[13] Gibson, G.R., Probert, H.M., Loo, J.V., et al. Dietary modulation of the human colonic microbiota: updating the concept of prebiotics. (2004) Nutr Res Rev 17(2): 259–275.
[14] Pompei, A., Cordisco, L., Raimondi, S., et al. In vitro comparison of the prebiotic effect of two inulin-type fructans. (2008) Anaerobe 14(5): 280–286.
[15] van de Wiele, T., Boon, N., Possemiers, S., et al. Inulin-type fructans of longer degree of polymerization exert more pronounced in vitro prebiotic effects. (2007) J Appl Microbiol 102(2): 452-460.
[16] Manach, C., Scalbert, A., Morand, C., et al. Polyphenols: food sources and bioavailability. (2004) Am J Clin Nutr 79(5): 727 - 747.
[17] Haslam, E. Plant polyphenols (syn. vegetable tannins) and chemical defense-A reappraisal. (1998) J Chem Ecol 14(10): 1789–1805.
[18] Scalbert, A. Antimicrobial properties of tannins. (1991) Phytochemistry 30(12): 3875–3883.
[19] Gu, Y.Y., Yu, S., Park, J.Y., et al. Dietary cocoa reduces metabolic endotoxemia and adipose tissue inflammation in high-fat fed mice. (2014) J Nutr Biochem 25(4): 439–445.
[20] Manach, C., Scalbert, A., Rémésy, C., et al. Polyphenols: Food sourcesand bioavailability. (2004) Am J Clin Nutr 79(5): 727–747.
[21] Brown, C.R. Antioxidants in potato. (2005) Am J Potato Res 82(2): 163–172.
Pubmed||Crossref||Others
[22] Hidalgo, M., Oruna-Concha, M.J., Kolida, S., et al. Metabolism of anthocyanins by human gut microflora and their influence on gut bacterial growth. (2012) J Agric Food Chem 60(15): 3882−3890.
[23] Jerzy, J., Joanna, M., Adam, J., et al. Consumption of polyphenol concentrate with dietary fructo-oligosaccharides enhances cecal metabolism of quercetin glycosides in rats. (2011) Nutrition 27(3): 351 –357.
[24] Zenon, Z., Jerzy, J., Isabel, E. Cecal parameters of rats fed diets containing grapefruit polyphenols and inulin as single supplements or in a combination. (2006) Nutrition 22(9): 898–904.
[25] Han, K.H., Sekikawa, M., Shimada, K., et al. Anthocyanin-rich purple potato flake extract has antioxidant capacity and improves antioxidant potential in rats. (2006) Br J Nutr 96(6): 1125 - 1134.
[26] Singleton, V.L., Orthofer, R., Lamuela-Raventós, R.M. Analysis of total polyphenol and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. (1998) Methods enzymol 299: 152-178.
[27] Han, K.H., Azuma, S., Fukushima, M. In vitro fermentation of spent turmeric powder with a mixed culture of pig faecal bacteria. (2014) Food Funct 5(10): 2446–2452.
[28] Scott, K.P., Duncan, S.H., Flint, H.J. Dietary fiber and the gut microbiota. (2008) Nutrition Bulletin 33(3): 201–211.
[29] Russell, W.R., Hoyles, L., Flint, H.J., et al. Colonic bacterial metabolites and human health. (2013) Curr Opin Microbiol 16(3): 246–254.
[30] Desai, M.S., Seekatz, A.M., Koropatkin, N.M., et al. A dietary fiber-deprived gut microbiota degrades the colonic mucus barrier and enhances pathogen susceptibility. (2016) Cell 167(5): 1339–1353.
[31] Sonnenburg, E.D., Smits, S.A., Tikhonov, M., et al. Diet-induced extinctions in the gut microbiota compound over generations. (2016) Nature 529(7585): 212–215.
[32] Koropatkin, N.M., Cameron, E.A., Martens, E.C. How glycan metabolism shapes the human gut microbiota. (2012) Nat Rev Microbiol 10(5): 323–335.
[33] Sonnenburg, E.D., Zheng, H.J., Joglekar, P., et al. Specificity of polysaccharide use in intestinal bacteroides species determines diet-induced microbiota alterations. (2010) Cell 141(7): 1241–1252.
[34] Cherbut C. Inulin and oligofructose in the dietary fibre concept. (2002) Br J Nutr 87: 159 – 162.
[35]Karimi, R., Azizi, M.H., Ghasemlou, M., et al. Application of inulin in cheese as prebiotic: fat replacer and texturizer: a review. (2015) Carbohydr Poly 119: 85–100.
[36] Roberfroid, M.B. Prebiotics and probiotics: are they functional foods? (2000) The American Journal of Clinical Nutrition 71(6): 1682-1687.
[37] Rastmanesh, R. High polyphenol, low probiotic diet for weight loss because of intestinal microbiota interaction. (2011) Chem-Biol Interact 189(1-2): 1 - 8.
[38] Parkar, S.G., Trower, T.M., Stevenson, D.E. Fecal microbial metabolism of polyphenols and its effect on human gut microbiota. (2013) Anaerobe 23: 12-19.
[39] Vendrame, S., Guglielmetti, S., Riso, P., et al. Six-week consumption of a wild blueberry powder drink increases Bifidobacteria in the human gut. (2011) J Agric Food Chem 59: 12815 – 21280.
[40] Tuohy, K.M., Conterno, L., Gasperotti, M., et al. Up-regulating the human intestinal microbiome using whole plant foods, polyphenolas and/or fiber. (2012) J Agric Food Chem 60(36): 8776–8782.
[41] Francesco, S., Lisa, C., Valentina, T., et al. Antagonistic effect of Lactobacillusstrains against gas-producing coliforms isolated from colicky infants. (2011) BMC Microbiology 11(1):157.
[42] Richard, K., Le, L., Ian, L.B., et al. A Synbiotic Combination of Resistant Starch and Bifidobacterium lactis Facilitates apoptotic Deletion of Carcinogen-Damaged Cells in Rat Colon. (2005) J Nutr 135(5): 996-1001.
[43] Canard, B., Cole, S.T. “Genome organization of the anaerobic pathogen Clostridium perfringens”. (1989) Proc Natl Acad Sci 86: 6676-6680.
[44] Lopez-Legarrea, P., Fuller, N., Zulet, M., et al. The influence of Mediterranean, carbohydrate and high protein diets on gut microbiota composition in the treatment of obesity and associated inflammatory state. (2014) Asia Pac J Clin Nutr 23(3): 360-368.
[45] Edwards, C.A., Rowland, I.R., Schweizer, T.F., et al. Dietary Fibre – A Component of Food. (1992) Springer Verlag 119-136.
Pubmed||Crossref||Others
[46] Scharlau, D., Borowicki, A., Habermann, N., et al. Mechanisms of primary cancer prevention by butyrate and other products formed during gut flora-mediated fermentation of dietary fibre. (2009) Mutat Res 682(1): 39-53.
[47] Fukuda, S., Toh, H., Hase, K., et al. Bifidobacteria can protect from enter pathogenic infection through production of acetate. (2011) Nature 469: 543-547.
[48] Tedelind, S., Westberg, F., Kjerrulf, M. Anti-inflammatory properties of the short-chain fatty acids acetate and propionate: a study with relevance to inflammatory bowel disease. (2007) World J Gastroenterol 13: 2826 -2832.
[49] Ishizaka, S., Kikuchi, E., Tsujii, T. Effect of acetate on the human immune system. (1993) Immunopharmacol Immunotoxicol 15(2-3): 151-162.
[50] Scheppach, W. Effects of short chain fatty acids on gut morphology and function. (1994) Gut 35(1): 35 - 38.
[51] Ding, H.H., Cui, S.W., Goff, H.D., et al. Short-chain fatty acid profiles from flaxseed dietary fibres after in vitro fermentation of pig colonic digesta: Structure–function relationship. (2015) Bioactive Carbohydrates and Dietary Fibre 6(2): 62–68.
[52] Venter, C.S., Vorster, H.H., Cummings, J.H. Effects of dietary propionate on carbohydrate and lipid metabolism in healthy volunteers. (1990) Am J Gastroenterol (5): 495-553.
[53] Baile, C.A. Metabolites as feedbacks for control of feed intake and receptor sites in goats and sheep. (1971) PhysiolBehav 7(1): 819–826.
[54] Anil, M.H., Forbes, J.M. Feeding in sheep during intraportal infusions of short-chain fatty acids and the effect of liver denervation. (1980) J Physiol 298: 407–414.
[55] Hinnebusch, B.F., Meng, S., Wu, J.T. The effects of short-chain fatty acids on human colon cancer cell phenotype are associated with histone hyperacetylation. (2002) J Nutr 132: 1012–1017.
[56] Han, K.H, Azuma, S., Fukushima, M. In vitro fermentation of spent turmeric powder with a mixed culture of pig faecal bacteria. (2014) Food and function 5: 2446-2452.
[57] Hague, A., Singh, B., Paraskeva, C. Butyrate acts as a survival factor for colonic epithelial cells: further fuel for the in vivo versus in vitro debate. (1997) Gastroenterology 112: 1036-1040.
[58] Pryde, S.E., Duncan, S.H., Hold, G.L. The microbiology of butyrate formation in the human colon. (2002) FEMS Microbiology Letters 217(2): 133-139.
[59] Perrin, P., Pierre, F., Patry, Y., et al. Only fibers promoting a stable butyrate producing colonic ecosystem decrease the rate of aberrant crypt foci in rats. (2001) Gut 48(1): 53-61.
[60] Rasmussen, H., Dirven, H.A.A.M., Grant, D., et al. Etiology of cecal and hepatic lesions in mice after administration of gas-carrier contrast agents used in ultrasound imaging. (2003) Toxicology and Applied Pharmacology 188(3): 176-184.
[61] Negi, P.S, Jayaprakasha, G.K. Antibacterial activity of grapefruit (Citrusparadisi) peel extract. (2001) Eur Food Res Technol 213(6): 484-487.
[62] Tebib, K., Besançon, P., Rouanet, J.M. Effects of dietary grape seed tannins on rat cecal fermentation and colonic bacterial enzymes. (1996) Nutr Res 6(1): 105–110.
[63] Davila Am., Blachier, F., Gotteland, M., et al. Intestinal luminal nitrogen metabolism: role of the gut microbiota and consequences for the host. (2013) Parmacol Res 68(1): 95-107.
[64] Mitsuoka, T. Significance of dietary modulation of intestinal flora and intestinal environment. (2000) Bioscience Microflora 19(1): 15-25.
[65] Vanhoof, K., De Schrijver, R. Catholic University of Leuven, Laboratory of Nutrition B 3001 Leuven. (1996) Belgium Nutrition Research 16: 1035-1039.
Pubmed||Crossref||Others