Therapeutic Effects of Bifidobacterium Longum Kacc 91563 on Dncb-Induced Atopic Dermatitis-Like Skin Lesions in Nc / Nga Mice
Chun-Ho Bae1, Joung-Woon Lee2, Hyun-Ji Park2, Ki-Il Nam2, Ji-Hyun Kim2, Jun-Seol Park2, Si-Hwan Song2
Affiliation
1Aram, Inc., Gyeonggi-do, Republic of Korea
2Chemon, Inc., Gyeonggi-do, Republic of Korea
Corresponding Author
Chul-Kyu Lee, Address: 147, Gwanggyo-ro, Yeongtong-gu, Suwon-si, Gyeonggi- do16229, Republic of Korea, Tel: +82-31-888-6645; E-mail: cklee@chemon.co.kr
Citation
Lee, C.K., et al. Therapeutic Effects of Bifidobacterium Longum Kacc 91563 on Dncb-Induced Atopic Dermatitis-Like Skin Lesions in Nc / Nga Mice. (2018) Int J Food Nutr Sci 5(1): 30- 37.
Copy rights
© 2018 Lee, C.K. This is an Open access article distributed under the terms of Creative Commons Attribution 4.0 International License.
Keywords
Scratching behavior; Atopic dermatitis; IgE; Inflammatory cytokines
Abstract
Atopic dermatitis (AD) is a chronic inflammatory skin disease characterized by severe itch. This study was designed to evaluate the therapeutic effects of oral administration of Bifidobacterium longum, KACC 91563 (BLK) on 2, 4-dinitrochlorobenzene (DNCB) - induced AD-like skin lesions in NC / Nga mice. Three doses of BLK (333, 1667, and 3334 mg / kg / day, 7 times a week) were orally administrated to mice with DNCB-induced AD. The therapeutic effect of BLK was assessed by measuring skin AD score, scratching behavior, serum IgE levels, as well as the expression of COX-2, TNF-α, IL-4, IL-10, and IL-12 by immunohistochemistry. AD score, scratching frequency, serum IgE level, COX-2, TNF-α, IL-4, IL-10, and IL-12 increased in mice with DNCB-induced AD. BLK alleviated AD-like symptoms such as skin severity of AD score and scratching behaviors. In addition, BLK reduced IgE, COX-2, and TNF-α, IL-4, IL-10, and IL-12 expression. These results suggest that oral administration of BLK has beneficial therapeutic effects for AD patients.
Introduction
Atopic dermatitis (AD), a chronic inflammatory skin disease associated with cutaneous hyper-reactivity, affects approximately 3% of infants, 10 – 20% of children, and 1 – 3% of adults worldwide[1]. AD is characterized by several common symptoms including itching, erythema, eczema skin lesions, chronic relapse, and pruritus. In AD, the skin becomes extremely itchy followed by severe scratching behavior, which induces the production of pro-inflammatory cytokines[2]. This in turn activates immune cells and initiates an inflammatory cycle of allergic dermatitis accompanied by erythema, keratosis, and scaling[3]. Adrenocorticosteroids and anti-histamine agents have been used for treating AD and have demonstrated favorable ameliorating effects. However, these treatments are linked to serious side effects[4]. Consequently, the beneficial effects of alternative therapies or natural products have been extensively investigated to ameliorate AD[5,6].
The NC / Nga mouse is the first reported spontaneously occurring AD model[7,8]. Skin changes that closely mimic human AD are induced in NC / Nga mice following exposure to various environmental aeroallergens[9]. It was reported that 2, 4-dinitrochlorobenzene (DNCB), an electrophilic and cytotoxic benzene derivative, induces stable clinical AD-like skin diseases in NC / Nga mice[10,11]. Skin changes in NC / Nga mice are evidenced by scratching behavior, followed by the rapid development of erythema, lichenification with edema, and hemorrhage[12]. Histological examinations have revealed hyperplasia and dense accumulation of eosinophils and mast cells in skin lesions[13]. Along with these skin changes, NC / Nga mice exhibit elevated levels of total serum IgE[7,8]. DNCB - induced contact hypersensitivity pathogenesis is predominantly the result of T cell mediated immune responses[9,14]. Therefore, DNCB-treated NC / Nga mice have been used as a valuable animal model for screening anti-AD agents[15,16].
Most Bifidobacterium are prevalent in the human colon, and B. longum strains often dominate the colonic microbiota of breast-fed infants[17,18]. Several studies suggest that B. longum provides potential health benefits to humans over a wide range of health conditions such as allergies, anti-inflammation, antibiotic-diarrhea, cholesterol reduction, and cancer prevention[19,20]. Recently, it was reported that B. longum KACC 91563 specifically induces apoptosis of mast cells and alleviates food allergy symptoms[21], and its casein hydrolysate is a potent antioxidant[22]. The present study was conducted to evaluate whether B. longum KACC 91563 can alleviate AD-like symptoms in DNCB-induced NC / Nga mice.
Materials and Methods
Animals
Male seven-week-old NC / Nga mice were purchased from SLC Inc. (Hamamatsu, Japan). This study was performed within the animal facility of the Gyeonggi Biocenter. The animals were housed in a room that was maintained at a temperature of 23 ± 3°C and relative humidity of 55 ± 15%, with a 12 - h light-dark cycle and 150 – 300 Lux luminous intensity. Throughout the experimental period, the temperature and humidity of the animal room were measured every hour with a computer-based automatic sensor. There were no major deviations in the temperature or humidity observed that could adversely affect experimental outcomes. Animals were offered irradiation-sterilized pellets as food (Teklad certified irradiated global 18% protein rodent diet; 2918 C, Envigo, UK). All animal experiments were performed based on the standard operating procedure of Chemon Inc. and the animal experimentation policy of the Gyeonggi Biocenter (experimentation number: 2016-10-0012).
B. longum KACC 91563 preparation
B. longum KACC 91563 (BLK) was provided by the National Institute of Animal Science, Rural Development Administration. The bacteria were isolated from the feces of healthy Korean infants. Mediogen, Inc (Seoul, Republic of Korea) manufactured the freeze-dried powder form of BLK, which includes 100 billion colony-forming units (cfu) / g. The minimal dose administered was one billion cfu / head / day (333 mg / kg / day); however, doses of 50 and 100 billion cfu / head / day were also tested.
Induction of atopic dermatitis-like symptoms
All mice were anesthetized using Zoletil, and then the dorsal hair was shaved using an electronic clipper. After 24 hrs, 200 μL of 1% DNCB (237329, Sigma-Aldrich, USA) in acetone / olive oil (3:1) was applied to the back skin, and a second application was performed after 3 days for sensitization. Seven days after the first application, 150 μL of 0.4% DNCB in acetone/olive oil (3:1) was applied to challenge the dorsal skin, three times a week for five weeks[23]. The guidelines presented by the Korean Food and Drug Administration (guideline No.1 – 019 – 2010 - 0000) for evaluating the efficacy of herbal medicine on atopic dermatitis were referred to for the induction of AD - like symptoms. Seven days after the first application, the mice were divided into six groups of 10 mice each. BLK (7 times / week) and dexamethasone (3 times / week) were then administrated for 5 weeks. The normal control group (Normal) received the vehicle (acetone / olive oil; 3:1) via oral administration. The atopic control group (DNCB) was treated with DNCB and administrated the vehicle. BLK- treated groups received DNCB and BLK at a dose of 333 mg / kg / day (DNCB + BLK 333), 1667 mg / kg / day (DNCB + BLK 1667), or 3334 mg / kg / day (DNCB + BLK 3334). The positive control group was topically treated with DNCB and dexamethasone at a concentration of 10 μM / head / day (DNCB + Dexa).
Skin clinical score
To evaluate any improvements in the condition of the skin following treatment with BLK, mice were anesthetized and photographed on the last day of the study, just before euthanasia. The atopic dermatitis score for skin lesions was measured once a week. The scores were graded from 0 to 3 (0: none, 1: mild, 2: moderate, 3: severe) for each of five symptoms: (i) erythema, (ii) dry skin, (iii) edema & hematoma, (iv) erosion, and (v) lichenification. A total dermatitis score, indicating clinical severity, was defined as the sum of all scores (maximum score: 15).
Measurement of scratching frequency
To evaluate scratching behavior, mice were placed into cages and the scratching frequency was assessed over a 30 - min period. Grooming behavior was excluded from the assessment of scratching frequency except during the last week.
Animal sacrifice and collection of samples
The animals were sacrificed by decapitation on the day of necropsy, and blood sampling was performed after anesthesia using isoflurane. Trunk blood was collected in a vacutainer tube containing clot activator, and centrifuged at 3000 rpm for 15 min. Serum samples were separated and stored at -80°C.
Morphological analysis of skin tissue
Skin samples were cut, fixed in 10% buffered formalin, and embedded in paraffin. After paraffin embedding, 3 – 4 μm serial sections were prepared. Representative sections were stained with hematoxylin and eosin (H&E) for general histopathology, Masson’s trichrome (MT) stain for collagen fiber, and Toluidine blue (TB) stain for mast cells, according to previously established methods[4,24,14,15]. The histological profiles of individual cross sections of the dorsal back skin were light microscopically observed (Model Eclipse 80i, Nikon, and Tokyo, Japan). Total thicknesses of the skin from epidermis to dermis (μm / skin), mean epithelial thickness (μm / skin), and the mean number of inflammatory cells infiltrating the dermis (cells / mm2 of dermis) were calculated for histomorphometrical analysis using a computer-assisted image analysis program (iSolution FL ver 9.1, IMT i-solution Inc., Vancouver, Quebec, Canada), according to previous methods with some modifications. H & E staining was used to assess infiltrating inflammatory cells, with mean mast cell numbers in the dermis (cells/mm2 of dermis) assessed by TB stain. Collagen fiber occupied regions in the hair follicle, excluding the dermis; (% / mm2 of dermis) were assessed by MT stain. At least five histological fields were evaluated in each prepared dorsal back skin, and histological sections were considered to calculate the mean histomorphometrical value.
Serum immunoglobulin E (IgE)
Quantitative measurement of serum IgE was performed using a commercial ELISA kit (AKRIE-010, Shibayagi, Japan), according to the manufactures protocol.
Immunohistochemistry
After necropsy, dorsal back skins were fixed in 10% neutral buffered formalin solution and the expression of inflammatory cytokines was evaluated by immune histo chemistry. Paraffin blocks of all dorsal back skins were serially sectioned into 3 – μm – thick sections, individually. Histological sections were prepared for all sample blocks, which were received as five serial sections in each block. The expression of cyclooxygenase - 2 (COX-2) (Cayman, An Arbour, MI, ISA), a key enzyme for the synthesis of prostaglandins, which are chemical mediators of inflammation[25,26] TNF-α (Santa Cruz Biotechnology, Santa Cruz, CA), a pro-inflammatory cytokine[27], and helper T cell related inflammatory cytokines – IL - 4 (Santa Cruz Biotechnology, Santa Cruz, CA), IL-10 (Abcam, Cambridge, UK), and IL 12[28] on the epidermis and / or dermis of dorsal back skin tissues was evaluated using purified primary antibodies with an ABC and peroxidase substrate kit (Vector Labs, Burlingame, CA, USA), respectively. Briefly, endogenous peroxidase activity was blocked by incubation in methanol and 0.3% H2O2 for 30 min. Non-specific binding of immunoglobulin was blocked with normal horse serum for 1 hr in a humidity chamber, after heating (95 – 100°C)-based epitope retrieval in 10 mM citrate buffer ( pH 6.0 ). Primary antibody were treated overnight at 4°C in the humidity chamber, and then incubated with biotinylated universal secondary antibody and ABC reagents for 1 hr at room temperature in the humidity chamber. Finally, sections were reacted with a peroxidase substrate for 3 min at room temperature. All sections were rinsed in 0.01M PBS three times between each step. Cells with over 20% immunoreactivity of key immune markers, against each antiserum compared to background were regarded as positive.
Statistical analysis
The data were analyzed using the SPSS statistics 22 for medical science by the LSD test method. The immunohistochemistry results were analyzed by LSD or Mann-Whitney test. Statistical significance was predefined as P ≤ 0.05 or 0.01.
Results and Discussion
BLK decreased symptoms of DNCB-induced atopic dermatitis
The atopic dermatitis-like symptoms in NC / Nga mice were induced by applying DNCB to the backs of mice for 7 weeks. To evaluate the effects of BLK on atopic dermatitis, BLK was orally administrated for 5 weeks. The experimental design is summarized in Figure 1. The skin clinical scores of the DNCB group significantly increased compared to that of the control group during the 5 - week study period. In addition, BLK treatment significantly impacted DNCB-induced atopic dermatitis in mice based on the skin clinical score (Figure 2). In the second week, BLK - treated and dexamethasone - treated mice showed a significant decrease in clinical scores (DNCB + BLK 333: 5.50 ± 0.19, DNCB + BLK 3334: 5.75 ± 0.31, DNCB + Dexa: 5.63 ± 0.26) compared to DNCB-treated mice (7.50 ± 0.53), with the exception of the DNCB + BLK 1667 group. In the third week, BLK - treated and dexamethasone - treated mice demonstrated a significant decrease in clinical scores (DNCB + BLK 333: 7.75 ± 0.59, DNCB + BLK 1667: 7.13 ± 0.35, DNCB + BLK 3334: 7.38 ± 0.32, DNCB + Dexa: 5.50 ± 0.50) compared to DNCB - treated mice (9.88 ± 0.64). In the fourth week, BLK - treated mice and dexamethasone - treated mice showed a significant decrease in clinical scores (DNCB + BLK 333: 8.63 ± 0.46, DNCB + BLK 1667: 8.13 ± 0.55, DNCB + BLK 3334: 8.25 ± 0.53, DNCB + Dexa: 6.38 ± 0.71) compared to DNCB - treated mice (10.63 ± 0.80). In the fifth week, there was a significant decrease in the clinical scores of BLK - and dexamethasone - treated mice (DNCB + BLK 333: 8.50 ± 0.46, DNCB + BLK 1667: 8.50 ± 0.50, DNCB + BLK 3334: 8.63 ± 0.53, DNCB + Dexa: 6.88 ± 0.88) compared to that of DNCB - treated mice (11.88 ± 0.58).
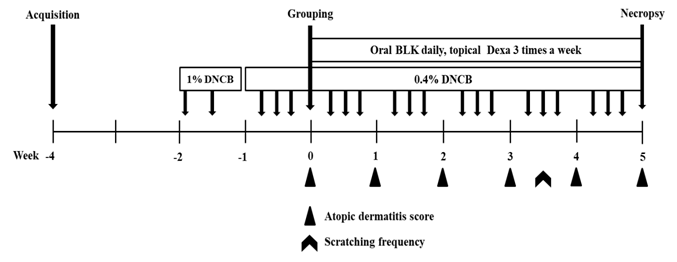
Figure 1: Experimental design. After one week of DNCB application on the dorsal skin of NC/Nga mice, daily oral administration of BLK or topical administration of dexamethasone was conducted for 5 weeks. During this time, DNCB was also administered as summarized in the experimental design.
Figure 2: Skin clinical scores in NC/Nga mice. (A) Photographs for clinical score measurement in week 1, 3, and on the day of euthanasia. (a) Normal, (b) DNCB, (c) DNCB+BLK 333, (d) DNCB+BLK 1667, (e) DNCB+BLK 3334, (f) DNCB+Dexa. (B) Skin clinical score. Data were statistically analyzed using LSD test methods and are expressed as the mean±S.E.M.*, **: statistically different from the DNCB group (P < 0.05, P < 0.01, respectively); ++: statistical difference between the control and DNCB groups (P < 0.01)
BLK decreased DNCB-induced scratch behavior
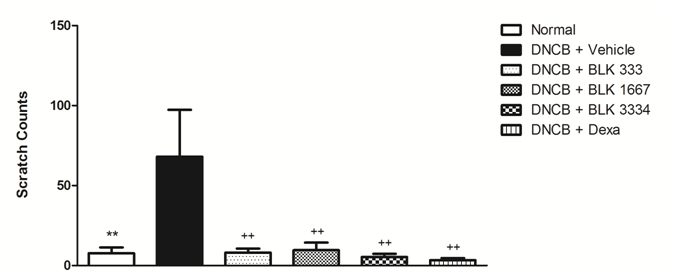
To assess the effects of BLK on scratching behavior, the number of scratches in the fourth week was measured (Figure 3). The number of scratches observed in the DNCB group (68.00 ± 29.35) was significantly higher than that observed in the control group (7.63 ± 3.66). In addition, the number of scratches following BLK treatment was attenuated in all BLK-treated groups (DNCB-BLK 333, 1667, and 3334: 8.00 ± 2.57, 9.57 ± 4.72, and 5.29 ± 2.00, respectively) compared to that in the DNCB group.
Figure 3: Evaluation of scratch behavior in NC/Nga mice. Values represent the scratching behavior in the fourth week. Data were statistically analyzed by box-plot and LSD test methods and are expressed as mean ± S.E.M.*, **: statistically different from the DNCB group (P < 0.05, P < 0.01, respectively); ++: statistical difference between the control and DNCB groups (P < 0.01)
BLK inhibited AD-induced increases in IgE
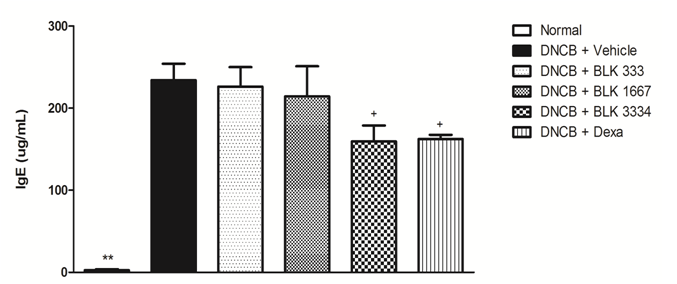
To investigate the effect of BLK on the DNCB - induced immune response, serum IgE level was determined (Figure 4). Serum IgE levels in the DNCB group (234.08 ± 19.90 μg / mL) were significantly increased compared to those in the control group (2.57 ± 1.23 μg / mL). Moreover, serum IgE in the DNCB + BLK 3334 group (159.48 ± 19.40 μg / mL) and the DNCB + Dexa group (162.24 ± 5.28 μg / mL) significantly decreased compared to that in the DNCB group.
Figure 4: IgE levels in the serum of NC/Nga mice. The quantitative measurement of serum IgE collected at necropsy. Data were statistically analyzed using LSD test methods and are expressed as the mean±S.E.M. *: statistically different from the DNCB group (P < 0.05); ++: statistical difference between the control and DNCB groups (P < 0.01).
BLK ameliorated histological atopic dermatitis
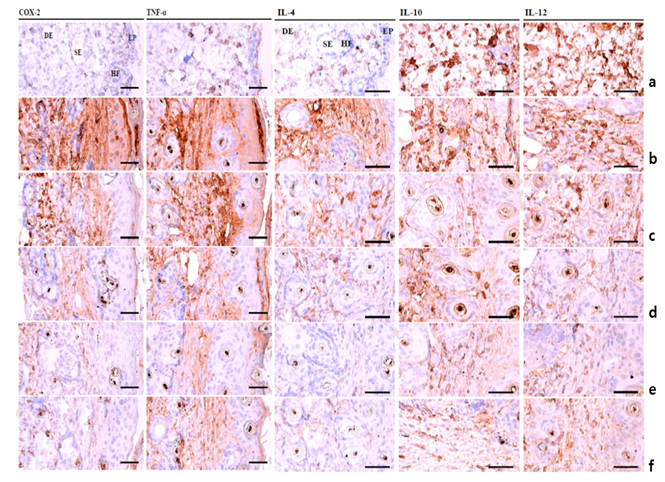
DNCB - induced atopic dermatitis mice showed significant increases in COX- 2 and TNF-α expression in both the epidermis and dermis.IL-4, IL-10, and IL-12expression in the dermis also increased compared to that in control mice, suggesting that DNCB stimulated hypersensitivity and accelerated the inflammatory process in allergic / atopic dermatitis mice. However, BLK significantly inhibited the DNCB stimulated increase in COX-2, TNF- α, IL- 4, IL-10, and IL-12 expression on the epidermis and / or dermis of dorsal back skin tissues in a dose-dependent manner (Table 1, Figure 5). All three different dosages of BLK showed inhibitory effects on the increased levels of COX-2, TNF-α, IL-4, IL-10, and IL - 12 in the epidermis and / or dermis of DNCB - treated AD dorsal back skins. These results demonstrate that oral treatment of BLK favorably suppresses skin inflammation by inhibiting the numerous DNCB-stimulated inflammatory responses, at dose levels of 333, 1667, and 3334 mg / kg / day.
Figure 5: Immunohistochemical profiles of dorsal skin tissues showing immunoreactivity. (a) Control, (b) DNCB, (c) DNCB+BLK 333, (d) DNCB+BLK 1667, (e) DNCB+BLK 3334, (f) DNCB+Dexa. Avidin-biotin complex-based immunohistochemical staining. Scale bars = 160 μm.*, **: statistically different from the DNCB group (P < 0.05, P < 0.01, respectively); ++: statistical difference between the control and DNCB groups (P < 0.01).
Table 1: Immunohistochemical analysis of dorsal back skin tissues in NC/Nga mice
| GROUP | Cyclooxygenase-2 (COX-2) | TNF-α | IL-4 | IL-10 | IL-12 | ||
|---|---|---|---|---|---|---|---|
| Epidermis (%) | Dermis (cells/mm2) | Epidermis (%) | Dermis (cells/mm2) | Dermis (cells/mm2) | Dermis (cells/mm2) | Dermis (cells/mm2) | |
| Normal | 7.25 ± 1.18 | 33.50 ± 6.32 | 4.38 ± 1.12 | 14.13 ± 2.48 | 3.75 ± 0.67 | 9.00 ± 1.28 | 6.63 ± 0.94 |
| DNCB | 70.75 ± 6.48++ | 542.50 ± 63.01++ | 65.63 ± 4.80++ | 774.00 ± 28.61++ | 163.50 ± 8.83++ | 324.50 ± 23.69++ | 169.75 ± 17.56++ |
| DNCB+BLK 333 | 36.50 ± 6.51** | 341.50 ± 35.10* | 31.88 ± 5.08** | 407.13 ± 39.81** | 110.75 ± 5.89** | 159.00 ± 23.53** | 92.00 ± 10.39** |
| DNCB+BLK 1667 | 21.00 ± 3.72** | 215.00 ± 31.91** | 26.63 ± 4.14** | 335.50 ± 49.28** | 80.25 ± 4.79** | 79.50 ± 12.63** | 65.00 ± 7.40** |
| DNCB+BLK 3334 | 19.25 ± 2.46** | 191.50 ± 32.57** | 22.00 ± 4.14** | 162.75 ± 31.88** | 53.00 ± 7.32** | 50.00 ± 4.28** | 50.00 ± 6.19** |
| DNCB+Dexa | 13.25 ± 2.80** | 140.75 ± 28.79** | 12.63 ± 1.38** | 118.50 ± 10.51** | 24.50 ± 3.50** | 19.00 ± 2.88** | 26.00 ± 2.93** |
Data were statistically analyzed by LSD and Mann-Whitney test methods and are expressed as the mean ± S.E.M. Normal: normal control; DNCB: DNCB-induced negative control; DNCB+BLK 333: DNCB-induction and BLK 333 mg/kg/day; DNCB+BLK 166: DNCB-induction and BLK 1667 mg/kg/day; DNCB+BLK 3334: DNCB-induction and BLK 3334 mg/kg/day: DNCB+Dexa: DNCB-induction and dexamethasone 10 μM.*, **: statistically different from the DNCB group (P < 0.05, P < 0.01, respectively); ++: statistical difference between thecontrol and DNCB groups(P < 0.01).
Conclusion
AD is a chronic inflammatory skin disease associated with immunological abnormalities and exposure to allergens[29]. Unfortunately, agents such as steroids and antihistamine drugs for AD therapy can have severe side effects that limit their clinical application. In this study, treatment with BLK in AD mice has been shown to significantly alleviate DNCB - induced dermatitis and scratching behavior. To our knowledge, this is the first study that illustrates the protective role of BLK in DNCB - induced AD mice.
Until date, two hypotheses concerning the causes of AD have been proposed. The first hypothesis suggests an immunological disturbance that leads to IgE - mediated sensitization, with epithelial barrier dysfunction[30]. The second implicates an intrinsic defect in the epithelial cells leading to barrier dysfunction[31].
In addition, TNF-α is an essential mediator of AD[32,33]. Therefore, the up regulation of total serum IgE and TNF-α levels are hallmarks of AD[34].
IgE expression is known to cause both acute and chronic phase skin inflammation. Clinical observations suggest a correlation between serum IgE and the extent of disease[35]. Patients suffering from AD are known to exhibit elevated serumlevels of total IgE[36]. IgE-mediated mast cell activation leads to a release of various kinds of chemical mediators. Chemical mediators facilitate the infiltration of inflammatory cells into the skin, such as eosinophils and lymphocytes[37]. The major causes of AD include increased serum IgE levels, eosinophilia, increased basophil spontaneous histamine release, activation of T lymphocyte, decrease cell-mediated immune response, and abnormality of the cytokine system. In addition, TNF-α is an essential mediator of AD[28,29]. Therefore, the upregulation of total serum IgE and TNF-α levels are hallmarks of AD[30].
Animal models for AD are based on repeated epicutaneous sensitization of tape stripped skin with ovalbumin or sensitization with hapten such as oxazolone and trinitrochlorobenzene. It is also possible to use IL-4, IL-18, and IL-31transgenic mouse models for studying AD. The NC / Nga mouse, NOA mouse, and DS - Ng mouse are spontaneous mouse models of AD[7]. In this study, we investigated the effects of oral treatment of BLK in DNCB-induced AD-like NC / Nga mice. DNCB-treated mice showed significant increases in AD score, scratching behavior, serum IgE levels, and the number of cells expressing COX-2, TNF-α, IL-4, IL-10, and IL-12 compared to control mice. In addition, BLK treatment attenuated these increases in the DNCB-treated group. Therefore, we conclude that BLK has the ability to significantly improve pruritus. Using immunohistochemistry, we evaluated the levels of COX-2, which is involved in angiogenesis and progression and the pathogenesis of AD[15]; TNF-α, a 17 - kDa protein, which was first identified as a product of activated macrophages and a well-known pro inflammatory cytokine that mediates the initiation of AD[38]; IL-4, a stimulator of type 2 helper T cell immune responses such as IgE switching of activated B cells; IL-10, a cytokine with multiple roles in immunoregulation and inflammation; and IL-12, which is naturally produced by dendritic cells, macrophages, neutrophils, and human B - lymphoblastoid cells in the response to antigenic stimulation[39]. We observed that BLK - treated mice showed significant decreases in COX-2, TNF-α, IL-4, IL-10, and IL-12 positive cells compared to DNCB - induced atopic dermatitis mice.
In conclusion, we have shown for the first time that BLK can ameliorate DNCB-induced AD-like symptoms via multiple mechanisms such as: (1) blocking AD-induced expression of IgE; (2) attenuating AD-induced increases in the levels of serum pro-inflammatory cytokines such as COX-2 and TNF-α; and (3) attenuating AD-induced reductions in the levels of serum anti-inflammatory cytokines such as IL-4, IL-6, and IL-10. Clinical trials of BLK in human patients will be needed. We believe that our experimental results can be readily translated to clinical use, as BLK is a natural food supplement and could easily be added to patient protocols.
Ethics statement: All animal experiments were performed based on the standard operating procedure of Chemon Inc. and the Animal Experimentation Policy of the Gyeonggi Biocenter, Gyeonggi do, Korea (experimentation number: 2016-10-0012).
Disclaimer: The respective individual authors related to this article are responsible for the all published data and any mistakes. The publisher does not take any such responsibilities.
References
- 1. Li, C., Lasse, S., Lee, P., et al. Development of atopic dermatitis-like skin disease from the chronic loss of epidermal caspase-8. (2010) Proc Natl Acad Sci U S A 107(51): 22249-22254.
- 2. Leung,D.Y. Atopic dermatitis:new insights and opportunities for therapeutic intervention. (2000) J Allergy Clin Immunol105(5): 860-876.
- 3. Thestrup-Pedersen, K. Clinical aspects of atopic dermatitis. (2000) Clin Exp Dermatol 25: 535-543.
- 4. Kim, C.G., Kang, M., Lee, Y. H., et al. Bathing Effects of Various Seawaters on Allergic (Atopic) Dermatitis-Like Skin Lesions Induced by 2,4-Dinitrochlorobenzene in Hairless Mice. (2015) Evid Based Complement Alternat Med: 25.
- 5. Kim, S. R., Choi, H. S., Seo, H. S.,et al. Topical application of herbal mixture extract inhibits ovalbumin- or 2,4-dinitrochlorobenzene-induced atopic dermatitis. (2012) Evid Based Complement Alternat Med: 25.
- 6. Merial-Kieny, C., Mengual, X., Guerrero, D., et al. Clinical efficacy of Avene hydrotherapy measured in a large cohort of more than 10,000 atopic or psoriatic patients. (2011) J Eur Acad Dermatol Venereol1: 30-34.
- 7. Jin,H., He,R., Oyoshi,M., et al. Animal models of atopic dermatitis. (2009) J Invest Dermatol 129(1): 31- 40.
- 8. Takeda, K., Gelfand,E.W., Mouse models of allergic diseases. (2009) Curr Opin Immunol 21: 660-665.
- 9. Kim,K.H., Choung, S.Y., . Oral administration of Vaccinium uliginosum L. extract alleviates DNCB-induced atopic dermatitis in NC/Nga mice. (2014) J Med Food 17(12): 1350-1360.
- 10. Fujii, Y., Takeuchi, H., Sakuma, S., et al. Characterization of a 2,4-dinitrochlorobenzene-induced chronic dermatitis model in rats. (2009) Skin Pharmacol Physiol 22(5): 240-247.
- 11. Jung,B.G., Cho, S.J., Koh, H. B., et al. Fermented Maesil (Prunus mume) with probiotics inhibits development of atopic dermatitis-like skin lesions in NC/Nga mice. (2010) Vet Dermatol21(2): 184-191.
- 12. Ikoma, A.. Analysis of the mechanism for the development of allergic skin inflammation and the application for its treatment: keratinocytes mechanisms and management of itch in atopic dermatitis.their pathogenic involvement. (2009) J Pharmacol Sci110(3): 260-264.
Pubmed||Crossref||Others
- 13. Hattori,K., Nishikawa, M.K., Watcharanurak, k., et al. Sustained exogenous expression of therapeutic levels of IFN-gamma ameliorates atopic dermatitis in NC/Nga mice via Th1 polarization. (2010) J Immunol 184(5): 2729-2735.
- 14. Zhang, E. Y., Chen,A.Y., Zhu,B.T. Mechanism of dinitrochlorobenzene-induced dermatitis in mice: role of specific antibodies in pathogenesis. (2009) PLoS One 4: e7703.
- 15. Kim,W. Y., Kim, H.G., Lee, H.W., et al. A Herbal Formula, Atofreellage, Ameliorates Atopic Dermatitis-Like Skin Lesions in an NC/Nga Mouse Model. (2015) Molecules21(1): E35.
- 16. Yang, G., An,D., Lee,M.H., et al. Choi Effect of Acer tegmentosum bark on atopic dermatitis-like skin lesions in NC/Nga mice. (2016) J Ethnopharmacol 177: 53-60.
- 17. Favier, C. F., de Vos, W. M., Akkermans, A. D. Development of bacterial and bifidobacterial communities in feces of newborn babies.(2003) Anaerobe9(5): 219-229.
- 18. Harmsen, H. J., Wildeboer-Veloo, A. C., Raangs, G. C., et al. Analysis of intestinal flora development in breast-fed and formula-fed infants by using molecular identification and detection methods. (2000) J Pediatr Gastroenterol Nutr 30(1): 61-67.
Pubmed||Crossref||Others
- 19. Oberreuther-Moschner, D. L., Jahreis, G., Rechkemmer,G.,et al. Dietary intervention with the probiotics Lactobacillus acidophilus 145 and Bifidobacterium longum 913 modulates the potential of human faecal water to induce damage in HT29clone19A cells. (2004) Br J Nutr 91: 925-932.
- 20. Xiao, J. Z., Kondo, S., Takahashi,N., et al. Hosono Effects of milk products fermented by Bifidobacterium longum on blood lipids in rats and healthy adult male volunteers. (2003) J Dairy Sci 86: 2452-2461.
- 21. Kim, J. H., Jeun, E.J., Hong, C. P., et al.. Extracellular vesicle-derived protein from Bifidobacterium longum alleviates food allergy through mast cell suppression. (2016) J Allergy ClinImmunol137(2): 507-516.
- 22. Chang,O.K., Seol,K.H., Jeong,S.G., etal.Casein hydrolysis by Bifidobacterium longum KACC91563 and antioxidant activities of peptides derived there from.(2013) JDairySci96(9): 5544-5555.
- 23. Park, S. J., Lee,H.A., Kim,J.W., et al. Platycodon grandiflorus alleviates DNCB-induced atopy-like dermatitis in NC/Nga mice. (2012) Indian J Pharmacol 44: 469-474.
- 24. Kim, K. H., Park, S. J., Lee, Y. J., et al. Inhibition of UVB-induced skin damage by exopolymers from Aureobasidium pullulans SM-2001 in hairless mice.(2015)BasicClinPharmacolToxicol116(2): 73-86.
- 25. Ling, H., Yang, H., Tan, S.H., et al. 6-Shogaol, an active constituent of ginger, inhibits breast cancer cell invasion by reducing matrix metalloproteinase-9 expression via blockade of nuclear factor-kappaB activation. (2010) Br J Pharmacol 161(8): 1763-1777.
- 26. Saba, E.,Lee, C.H., Jeong da, H., et al. Fermented rice bran prevents atopic dermatitis in DNCB-treated NC/Nga mice. (2016) J Biomed Res 30: 334-343.
- 27. , C. W., Park, S. M., Kim, Y. S., . Biomolecular evidence of anti-inflammatory effects by Clematis mandshurica Ruprecht root extract in rodent cells. (2014) J Ethnopharmacol 155(2): 1141-1155.
- 28. Choi, Y.Y., Kim, M. H., Lee, H., et al. Cynanchum atratum inhibits the development of atopic dermatitis in 2,4-dinitrochlorobenzene-induced mice. (2017) Biomed Pharmacother90: 321-327.
- 29. Boguniewicz, M., and. Leung, D.Y. Atopic dermatitis: a disease of altered skin barrier and immune dysregulation. (2011) Immunol Rev242(1): 233-246.
- 30. Sivaranjani, N., Rao,S.V., Rajeev,G. Role of reactive oxygen species and antioxidants in atopic dermatitis. (2013) J Clin Diagn Res 7: 2683-2685.
- 31. Bieber,T. Atopic dermatitis. (2008) N Engl J Med 358(14):1483-1494.
- 32. Auriemma, M., Vianale, G., Amerio, P. et al. Cytokines and T cells in atopic dermatitis.(2013) Eur Cytokine Netw24(1): 37-44.
Pubmed||Crossref||Others
- 33. Behniafard, N., Gharagozlou, M., Farhadi, E., et al.TNF-alpha single nucleotide polymorphisms in atopic dermatitis.(2012) Eur. Cytokine Netw 23(4): 163-165.
- 34. Arshad, S.H., Holgate,S. The role of IgE in allergen-induced inflammation and the potential for intervention with a humanized monoclonal anti-IgE antibody. (2001) Clin Exp Allergy31(9):1344-1351.
- 35. Peroni, D. G., Bodini,A., Corradi,M., et al. Markers of oxidative stress are increased in exhaled breath condensates of children with atopic dermatitis. (2012) Br J Dermatol 166: 839-843.
- 36. Ott, H., Stanzel,S., Ocklenburg,C.,et al. Total serum IgE as a parameter to differentiate between intrinsic and extrinsic atopic dermatitis in children. (2009) Acta Derm Venereol 89: 257-261.
- 37. Matsuda,H., Watanabe,N., Geba,G.P., et al.. Development of atopic dermatitis-like skin lesion with IgE hyperproduction in NC/Nga mice. (1997) Int Immunol 9(3): 461-466.
- 38. Coondoo, A.The role of cytokines in the pathomechanism of cutaneous disorders. (2012) Indian J Dermatol 57(2): 90-96.
- 39. Kalinski, P., Hilkens,C.M., Snijders, A., et al. IL-12-deficient dendritic cells, generated in the presence of prostaglandin E2, promote type 2 cytokine production in maturing human naive T helper cells. (1997) J Immunol 159(1): 28-35.